
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3.SE, Problem 25AP
Interpretation Introduction
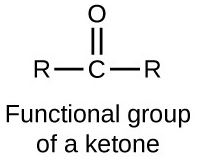
a)
Interpretation:
The hybridisation of the following molecules is to be shown with the help of diagram.
Concept introduction:
a)
Interpretation Introduction
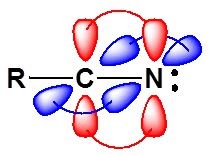
b)
Interpretation:
The hybridisation of the following molecules is to be shown with the help of diagram.
Concept introduction:
b)
Interpretation Introduction
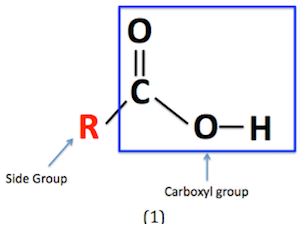
c)
Interpretation:
The hybridisation of the following molecules is to be shown with the help of diagram.
Concept introduction:
c)
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
(a) Explain bonding of methanol in terms of hybridization.
Compounds that contain an N-H group associate by hydrogen bonding.
(a) Do you expect this association to be stronger or weaker than that of compounds containing an O-H group?
(b) Based on your answer to part (a), which would you predict to have the higher boiling point, 1-butanol or 1-butanamine?
Describe how would you distinguish the following pairs,
(a) Benzene and cyclohexane
(b) Phenol and toluene
(c) Phenol and benzoic acid
(d) methanol and isopropyl alcohol
Chapter 3 Solutions
Organic Chemistry
Ch. 3.1 - Prob. 1PCh. 3.1 - Prob. 2PCh. 3.1 - Identify the functional groups in the following...Ch. 3.2 - Draw structures of the five isomers of C6H14.Ch. 3.2 - Propose structures that meet the following...Ch. 3.2 - Prob. 6PCh. 3.3 - Draw the eight 5-carbon alkyl groups (pentyl...Ch. 3.3 - Identify the carbon atoms in the following...Ch. 3.3 - Prob. 9PCh. 3.3 - Prob. 10P
Ch. 3.4 - Give IUPAC names for the following compounds:Ch. 3.4 - Prob. 12PCh. 3.4 - Name the eight 5-carbon alkyl groups you drew in...Ch. 3.4 - Give the IUPAC name for the following hydrocarbon,...Ch. 3.7 - Make a graph of potential energy versus angle of...Ch. 3.7 - Sight along the C2-C1 bond of 2-methylpropane...Ch. 3.7 - Sight along the C2-C3 bond of 2,3-dimethylbutane,...Ch. 3.7 - Draw a Newman projection along the C2-C3 bond of...Ch. 3.SE - Prob. 19VCCh. 3.SE - Prob. 20VCCh. 3.SE - Draw a Newman projection along the C2-C3 bond of...Ch. 3.SE - Prob. 22APCh. 3.SE - Prob. 23APCh. 3.SE - Propose structures for the following: (a) A...Ch. 3.SE - Prob. 25APCh. 3.SE - Draw the structures of the following molecules:...Ch. 3.SE - Draw structures that meet the following...Ch. 3.SE - Prob. 28APCh. 3.SE - In each of the following sets, which structures...Ch. 3.SE - There are seven constitutional isomers with the...Ch. 3.SE - Prob. 31APCh. 3.SE - Draw compounds that contain the following: (a) A...Ch. 3.SE - Prob. 33APCh. 3.SE - Draw and name all monochloro derivatives of...Ch. 3.SE - Draw structures for the following: (a)...Ch. 3.SE - Prob. 36APCh. 3.SE - Draw a compound that: (a) Has nine primary...Ch. 3.SE - Give IUPAC names for the following compounds:Ch. 3.SE - Name the five isomers of C6H14.Ch. 3.SE - Explain why each of the following names is...Ch. 3.SE - Prob. 41APCh. 3.SE - Consider 2-methylbutane (isopentane). Sighting...Ch. 3.SE - What are the relative energies of the three...Ch. 3.SE - Construct a qualitative potential-energy diagram...Ch. 3.SE - Prob. 45APCh. 3.SE - Draw the most stable conformation of pentane,...Ch. 3.SE - Draw the most stable conformation of...Ch. 3.SE - Prob. 48APCh. 3.SE - Prob. 49APCh. 3.SE - Formaldehyde, H2C=O, is known to all biologists...Ch. 3.SE - Prob. 51APCh. 3.SE - Increased substitution around a bond leads to...Ch. 3.SE - Prob. 53APCh. 3.SE - In the next chapter we'll look at...Ch. 3.SE - We’ll see in the next chapter that there are two...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2) Provide structures for the following compounds: a) ethyl acetate b) Pentanoic acidarrow_forward(4) Write the structure of the following compounds from their IUPAC names. (a) ethanamide (b) methylethanoate (c) propanoic acid (d) 2-butanone (5) Write the structure of the following ethers and amines from their common names (a) methyl propyl (b) trimethylamine (6) Decide whether the following alcohols are polar or nonpolar (a) CH3CH2CH2CH2CH2CH2CH2OH (b) CH3CH2OHarrow_forwardTRUE OR FALSE (a) There are three amines with the molecular formula C3H9N. (b) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (c) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (d) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (e) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (f) The molecular formula of the smallest carboxylic acid is C2H4O2.arrow_forward
- (a) Write the molecular formula of acetaminophen (see Fig. 7.34b).(b) A tablet of Extra Strength Tylenol contains 500 mg acetaminophen. Calculate the chemical amount (in moles) of that compound in the tablet.arrow_forwardIdentify which of the statements is/are correct. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic acid is C2H4O2.arrow_forwardWrite the chemical equation for a pentanoate ion acting as base when it reacts with hydrochloric acid (HCI). Which is formula of this rule of reaction?arrow_forward
- Describe concisely a chemical test to distinguish between the following pairs of compounds.(a) Propanal and propanone(b) Phenol and benzoic acid(c) Hexan-3-one and hexan-2-onearrow_forward(i) Draw the structure of any amine and give the IUPAC name of that amine. (i) Classify the amine in your answer provided in (i) above (iii) Draw the structure of ethyl butanoate and name the functional group. (iv) Give the IUPAC name of the following compound and name the functional group:arrow_forwardIdentify the class of the compound based on its functional groups. .0. H3C CH3 H3C° H3C H. (a) (b) (c) alcohol alcohol alcohol ether ether ether aldehyde aldehyde aldehyde ketone ketone ketone carboxylic acid carboxylic acid carboxylic acid ester ester ester amine amine amine :0:arrow_forward
- Give a chemical test to distinguish between each of the following pairs of compounds :(i) Ethylamine and Aniline(ii) Aniline and Benzylaminearrow_forward(a) Compound Z is a tertiary aromatic amine with the formula, C8H11N. Provide a chemical structure for compound Z. (b)nDraw the structure of the product formed exclusively when nitrous acid reacts with Z.arrow_forwardIn each of the following reactions, two possible organic products can be formed. Draw both organic products in each case and then circle the one formed in greatest quantity in each case. HC (a) 1) NaH, 2) acid (b) CH,CH,OH (c) CH,CH,OH NH2 (d) Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning