
Concept explainers
(a)
Interpretation:
It is to be determined whether the two given molecules are constitutional isomers of each other.
Concept introduction:
The compounds having same molecular formula but different connectivity of the atoms are constitutional isomers. The arrangement of the atoms is different for constitutional isomers. From the total number of atoms and arrangement of atoms, it is decided that whether the two molecules are constitutional isomers or not.
Answer to Problem 4.16P
The two given molecules are not constitutional isomers of each other.
Explanation of Solution
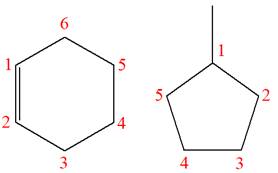
The structures of the given molecules are
The molecular formula of the first compound is
The first molecule contains ten hydrogens, and the second molecule contains twelve hydrogens. However, the total number of hydrogens is different in each molecule. In case of constitutional isomers, the total number of atoms remains the same in the molecular formula. So both molecules are not constitutional isomers of each other.
The constitutional isomers are determined from the molecular formula and connectivity of the atoms.
(b)
Interpretation:
It is to be determined whether the two given molecules are constitutional isomers of each other.
Concept introduction:
The compounds having same molecular formula but different connectivity of the atoms are constitutional isomers. The arrangement of the atoms is different for constitutional isomers. From the total number of atoms and arrangement of atoms, it is decided that whether the two molecules are constitutional isomers or not.
Answer to Problem 4.16P
The two given molecules are not constitutional isomers of each other.
Explanation of Solution
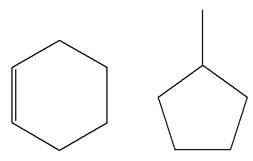
The structures of the given molecules are
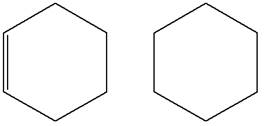
The molecular formula of the first compound is
The first molecule contains ten hydrogens, and the second molecule contains twelve hydrogens. However, the total number of hydrogens is different in each molecule. In case of constitutional isomers, the total number of atoms remains the same in the molecular formula. So both molecules are not constitutional isomers of each other.
The constitutional isomers are determined from the molecular formula and connectivity of the atoms.
(c)
Interpretation:
It is to be determined whether the two given molecules are constitutional isomers of each other.
Concept introduction:
The compounds having same molecular formula but different connectivity of the atoms are constitutional isomers. The arrangement of the atoms is different for constitutional isomers. From the total number of atoms and arrangement of atoms, it is decided that whether the two molecules are constitutional isomers or not.
Answer to Problem 4.16P
The two given molecules are not constitutional isomers of each other.
Explanation of Solution
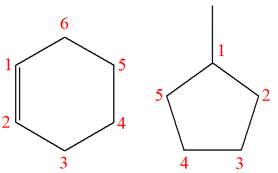
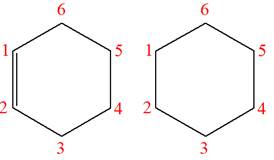
The structures of the given molecules are
The molecular formula of the first compound is

The first molecule contains ten hydrogens, and the second molecule contains twelve hydrogens. However, the total number of hydrogens is different in each molecule. In case of constitutional isomers, the total number of atoms remains the same in the molecular formula. So both molecules are not constitutional isomers of each other.
The constitutional isomers are determined from the molecular formula and connectivity of the atoms.
(d)
Interpretation:
It is to be determined whether the two given molecules are constitutional isomers of each other.
Concept introduction:
The compounds having same molecular formula but different connectivity of the atoms are constitutional isomers. The arrangement of the atoms is different for constitutional isomers. From the total number of atoms and arrangement of atoms, it is decided that whether the two molecules are constitutional isomers or not.
Answer to Problem 4.16P
The two given molecules are constitutional isomers of each other.
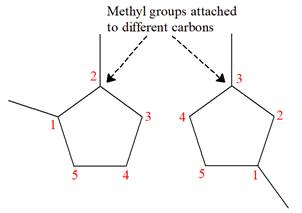
Explanation of Solution
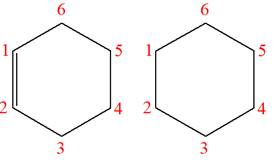
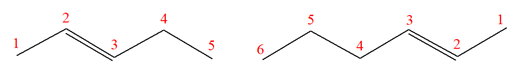
The structures of the given molecules are
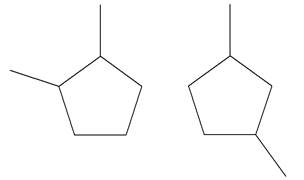
The molecular formula of both molecules is
The number assigned to carbon atoms are same in each molecule based on the first methyl group attached to the ring. However, the connectivity of the methyl group is not the same. In the first molecule, the methyl group is attached to the
The constitutional isomers are determined from the molecular formula and connectivity of the atoms.
(e)
Interpretation:
It is to be determined whether the two given molecules are constitutional isomers of each other.
Concept introduction:
The compounds having same molecular formula but different connectivity of the atoms are constitutional isomers. The arrangement of the atoms is different for constitutional isomers. From the total number of atoms and arrangement of atoms, it is decided that whether the two molecules are constitutional isomers or not.
Answer to Problem 4.16P
The two given molecules are not constitutional isomers of each other.
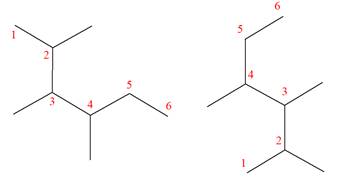
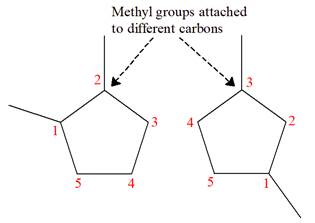
Explanation of Solution
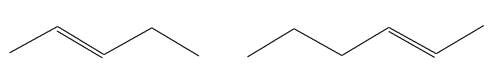
The structures of the given molecules are
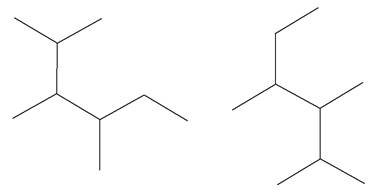
The molecular formula of both molecules is
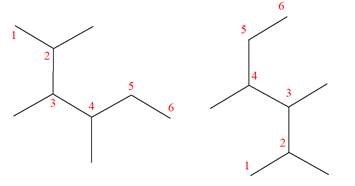
The numbers assigned to carbon atoms are the same in each molecule based on the first methyl group attached to the longest carbon chain. Both molecules have methyl groups attached to the
The constitutional isomers are determined from the molecular formula and connectivity of the atoms.
(f)
Interpretation:
It is to be determined whether the two given molecules are constitutional isomers of each other.
Concept introduction:
The compounds having same molecular formula but different connectivity of the atoms are constitutional isomers. The arrangement of the atoms is different for constitutional isomers. From the total number of atoms and arrangement of atoms, it is decided that whether the two molecules are constitutional isomers or not.
Answer to Problem 4.16P
The two given molecules are not constitutional isomers of each other.
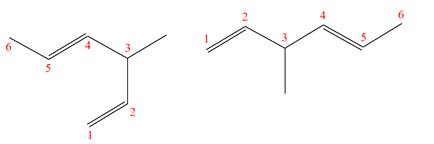
Explanation of Solution
The structures of the given molecules are
The molecular formula of both molecules is

The numbers assigned to double-bonded carbon atoms are the same in each molecule. In both compounds, the methyl group is connected to
The constitutional isomers are determined from the molecular formula and connectivity of the atoms.
(g)
Interpretation:
It is to be determined whether the two given molecules are constitutional isomers of each other.
Concept introduction:
The compounds having same molecular formula but different connectivity of the atoms are constitutional isomers. The arrangement of the atoms is different for constitutional isomers. From the total number of atoms and arrangement of atoms, it is decided that whether the two molecules are constitutional isomers or not.
Answer to Problem 4.16P
The two given molecules are not constitutional isomers of each other.
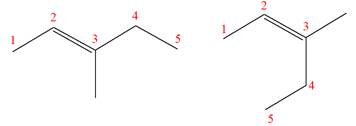
Explanation of Solution
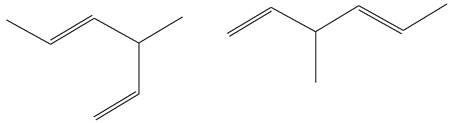
The structures of the given molecules are
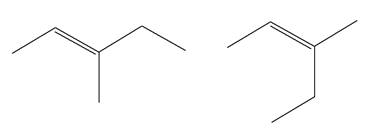
The molecular formula of both molecules is
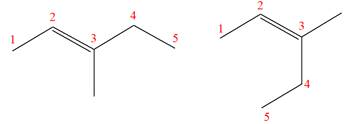
The number assigned to double-bonded carbon atoms are the same in each molecule. In both compounds, the methyl group is connected to
The constitutional isomers are determined from the molecular formula and connectivity of the atoms.
(h)
Interpretation:
It is to be determined whether the two given molecules are constitutional isomers of each other.
Concept introduction:
The compounds having same molecular formula but different connectivity of the atoms are constitutional isomers. The arrangement of the atoms is different for constitutional isomers. From the total number of atoms and arrangement of atoms, it is decided that whether the two molecules are constitutional isomers or not.
Answer to Problem 4.16P
The two given molecules are not constitutional isomers of each other.
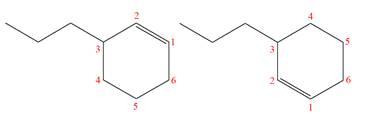
Explanation of Solution
The structures of the given molecules are
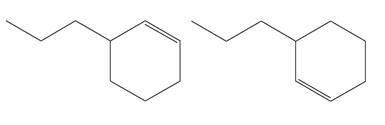
The molecular formula of both molecules is
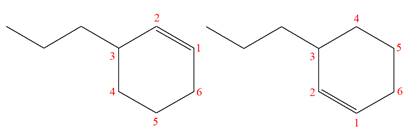
The numbers assigned to double-bonded carbon atoms are the same in each molecule. In both compounds, the propyl group is connected to
The constitutional isomers are determined from the molecular formula and connectivity of the atoms.
(i)
Interpretation:
It is to be determined whether the two given molecules are constitutional isomers of each other.
Concept introduction:
The compounds having same molecular formula but different connectivity of the atoms are constitutional isomers. The arrangement of the atoms is different for constitutional isomers. From the total number of atoms and arrangement of atoms, it is decided that whether the two molecules are constitutional isomers or not.
Answer to Problem 4.16P
The two given molecules are constitutional isomers of each other.
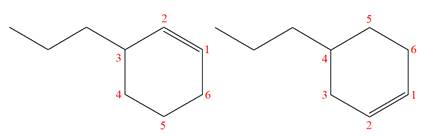
Explanation of Solution
The structures of the given molecules are
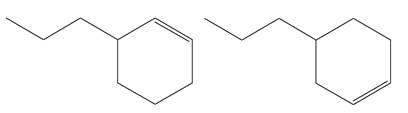
The molecular formula of both molecules is
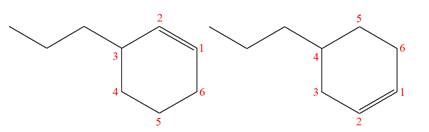
The numbers assigned to double-bonded carbon atoms are the same in each molecule.
However, the connectivity of the propyl group is not the same. In the first molecule, the propyl group is attached to the
The constitutional isomers are determined from the molecular formula and connectivity of the atoms.
Want to see more full solutions like this?
Chapter 4 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Check the box under each molecule in the table below that is an isomer of this molecule: If there are no isomers, check the none of the above box under the table. none of the abovearrow_forwardBe sure to answer all parts. Below is an incomplete list of all the constitutional isomers with the molecular formula C,H1,0. Draw the missing constitutional isomers and identify the functional group for each. CH3- CH2- CH2- CH2 - OH Functional Group: (select) CH2 — ОН CH3-C- CH3 Functional Group: (select) H. Functional Group: Alcohol draw structure ... CH3- CH2-0- CH2 - CH3 Functional Group: (select) Functional Group: Ether draw structurearrow_forwardIdentify the different functional groups in the following molecules using names from the table below. (If there are fewer than 3 different functional groups, leave an appropriate number of answer boxes empty.)arrow_forward
- Homework problem wants to different structures that have the same molecular formula but different connectivities for c5h12arrow_forwardA. For each of the following molecules, identify and label all of the functional groups present in the molecule. Then, draw in the lone pairs on the heteroatoms (any atom that is not carbon or hydrogen) that are assumed in each structure.arrow_forwardName both structures by writing the desired isomer pair of molecules given in each of the below.arrow_forward
- Provide the common name for the condensed formula shown herearrow_forwardPlease solve, with explanation. Need only 4.18 determine whether the following compounds are constitutional isomersarrow_forwardNOTE: In order to determine the identical compounds, name all the compounds. For the constitutional isomers, get the Molecular formula of each compound.arrow_forward
- Draw the constitutional isomers of the following molecular formulas.arrow_forwardHello, can you draw and name two different molecules (with the explicit formula) with a total of 12 carbons, together with the side groups, according to the options given? a)Write and name two cyclic amide molecule. b)Write and name two aromatic amine molecule.arrow_forwardWhy cycloethane and cyclomethane do not exist, whereas, cyclopropane does exist.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
