
Concept explainers
Energy exists in different forms in our natural world. In Figure 5.10, identify where:
- a. Potential (stored) energy of the fuel is converted to heat.
- b. Kinetic energy of water molecules is converted to mechanical energy.
- c. Mechanical energy is converted to electrical energy.
- d. Electrical energy is converted into forms such as heat and light.
(a)
Interpretation:
Energy exists in different forms in our natural world. From the given figure of components of an electric power plant it has to be identified that where potential energy of the fuel is converted to heat.
Concept Introduction:
A fuel is a substance which releases energy in the form of heat while undergoing combustion. Hydrocarbons which have high heats of combustion make it a best fuel.
The general equation for the complete combustion of a hydrocarbon is written below,
Coal is a good fuel becouse it is completely made up of carbon atoms. It is a fossil fuel.
Coals can be classified and graded according to the carbon percent and the impurity percent such as sulfur.
There different types of energy forms in the world and some of these energy forms are mentioned below,
Mechanical energy: Energy produced by a machine or a moving part.
Thermal energy: Energy produced by heat.
Light energy: Energy produced from light.
Potential energy: Energy stored in an object due to its position above the earth’s surface.
Kinetic energy: Energy produced by motion of an object.
Explanation of Solution
The given figure which depicts the components of an electric power plant is shown below,
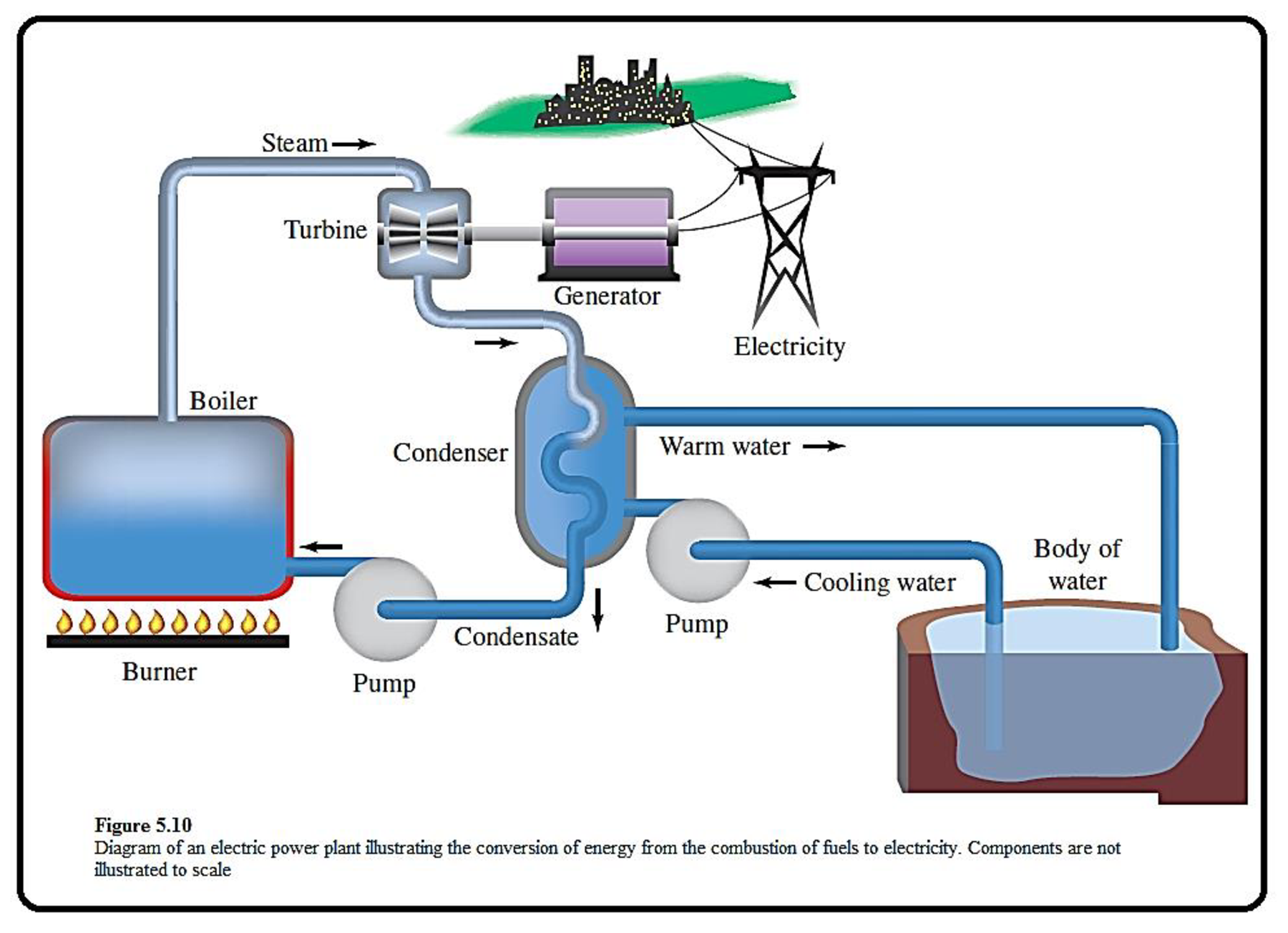
Figure 1
Coal is a best fuel to produce heat energy, the steam produced through this process is used to generate electricity.
Energy exists in different forms in our world.
In the given representation, the fuel in the burner is a source of potential energy. When it burned, some of its potential energy is converted to heat throgh combustion. This heat is converted into kinetic energy of steams.
(b)
Interpretation:
Energy exists in different forms in our natural world. From the given figure of components of an electric power plant it has to be identified that where kinetic energy of water molecule is converted to mechanical energy.
Concept Introduction:
A fuel is a substance which releases energy in the form of heat while undergoing combustion. Hydrocarbons which have high heats of combustion make it a best fuel.
The general equation for the complete combustion of a hydrocarbon is written below,
Coal is a good fuel becouse it is completely made up of carbon atoms. It is a fossil fuel.
Coals can be classified and graded according to the carbon percent and the impurity percent such as sulfur.
There different types of energy forms in the world and some of these energy forms are mentioned below,
Mechanical energy: Energy produced by a machine or a moving part.
Thermal energy: Energy produced by heat.
Light energy: Energy produced from light.
Potential energy: Energy stored in an object due to its position above the earth’s surface.
Kinetic energy: Energy produced by motion of an object.
Explanation of Solution
The given figure which depicts the components of an electric power plant is shown below,
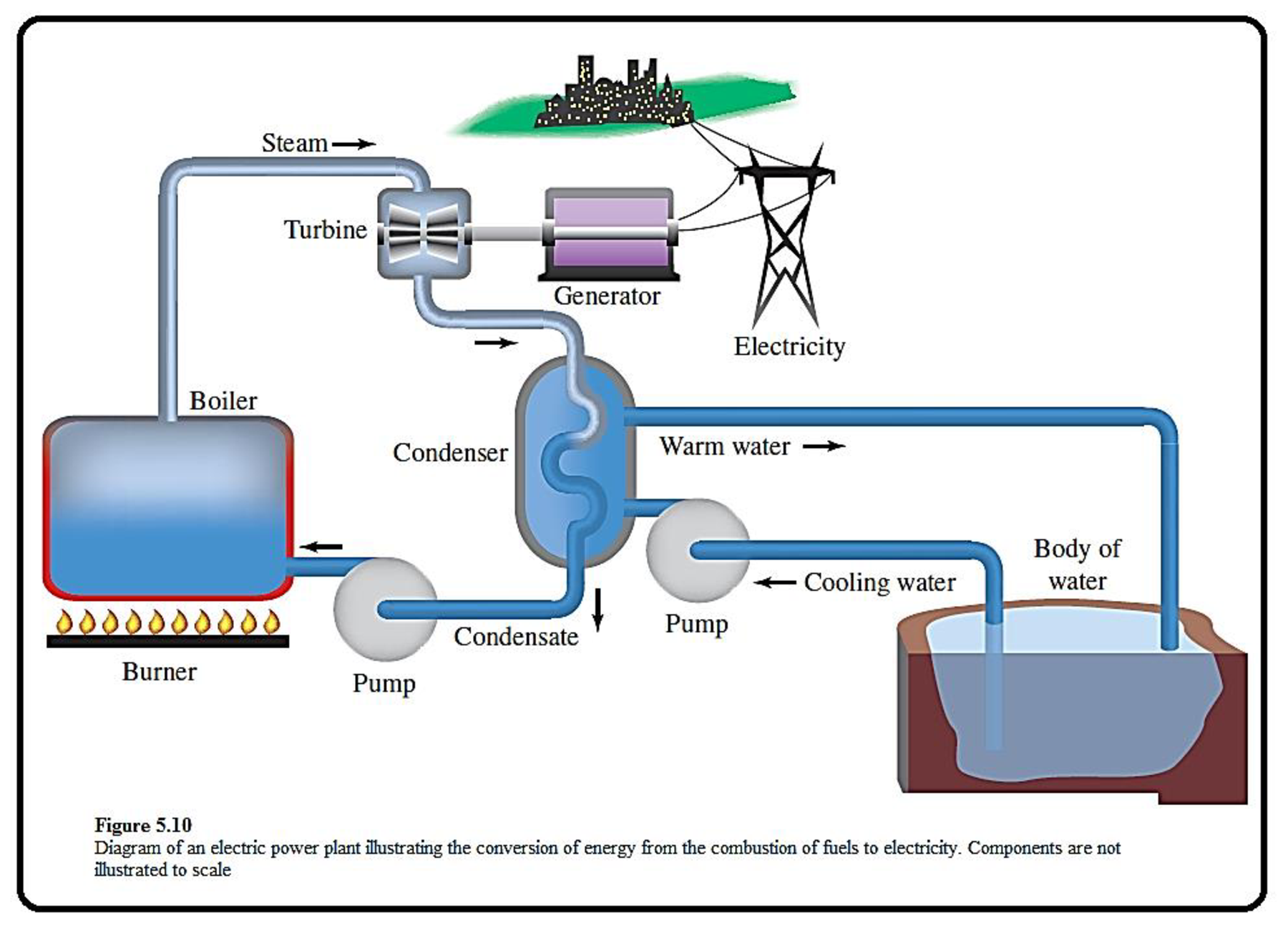
Figure 1
Coal is a best fuel to produce heat energy, the steam produced through this process is used to generate electricity.
Energy exists in different forms in our world.
In the given representation, the fuel in the burner is a source of potential energy. When it burned, some of its potential energy is converted to heat throgh combustion. This heat is converted into kinetic energy of steams. By spinning the turbine, the kinetic energy of the steam is converted to mechanical energy.
(c)
Interpretation:
Energy exists in different forms in our world. From the given figure of components of an electric power plant it has to be identified that where mechanical energy is converted to electrical energy.
Concept Introduction:
A fuel is a substance which releases energy in the form of heat while undergoing combustion. Hydrocarbons which have high heats of combustion make it a best fuel.
The general equation for the complete combustion of a hydrocarbon is written below,
Coal is a good fuel becouse it is completely made up of carbon atoms. It is a fossil fuel.
Coals can be classified and graded according to the carbon percent and the impurity percent such as sulfur.
There different types of energy forms in the world and some of these energy forms are mentioned below,
Mechanical energy: Energy produced by a machine or a moving part.
Thermal energy: Energy produced by heat.
Light energy: Energy produced from light.
Potential energy: Energy stored in an object due to its position above the earth’s surface.
Kinetic energy: Energy produced by motion of an object.
Explanation of Solution
The given figure which depicts the components of an electric power plant is shown below,
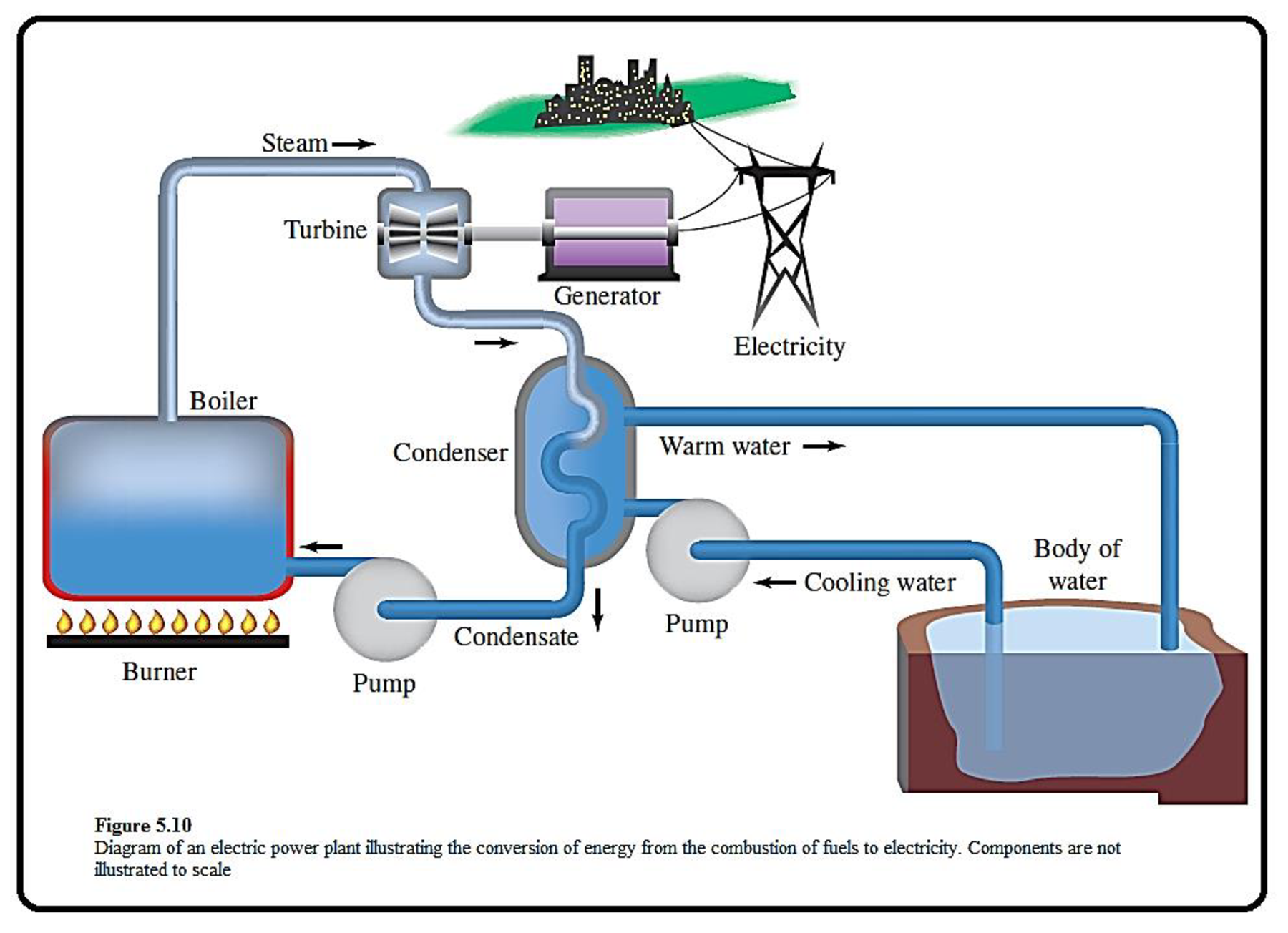
Figure 1
Coal is a best fuel to produce heat energy, the steam produced through this process is used to generate electricity.
Energy exists in different forms in our world.
In the given representation, the fuel in the burner is a source of potential energy. When it burned, some of its potential energy is converted to heat throgh combustion. This heat is converted into kinetic energy of steams. By spinning the turbine, the kinetic energy of the steam is converted to mechanical energy. The mechanical energy generated from the spinning turbne is converted to electrical energy by rotating a wire in a magnetic field.
(d)
Interpretation:
Energy exists in different forms in our world. From the given figure of components of an electric power plant it has to be identified that where electrical energy is converted into forms such as heat and light.
Concept Introduction:
A fuel is a substance which releases energy in the form of heat while undergoing combustion. Hydrocarbons which have high heats of combustion make it a best fuel.
The general equation for the complete combustion of a hydrocarbon is written below,
Coal is a good fuel becouse it is completely made up of carbon atoms. It is a fossil fuel.
Coals can be classified and graded according to the carbon percent and the impurity percent such as sulfur.
There different types of energy forms in the world and some of these energy forms are mentioned below,
Mechanical energy: Energy produced by a machine or a moving part.
Thermal energy: Energy produced by heat.
Light energy: Energy produced from light.
Potential energy: Energy stored in an object due to its position above the earth’s surface.
Kinetic energy: Energy produced by motion of an object.
Explanation of Solution
The given figure which depicts the components of an electric power plant is shown below,
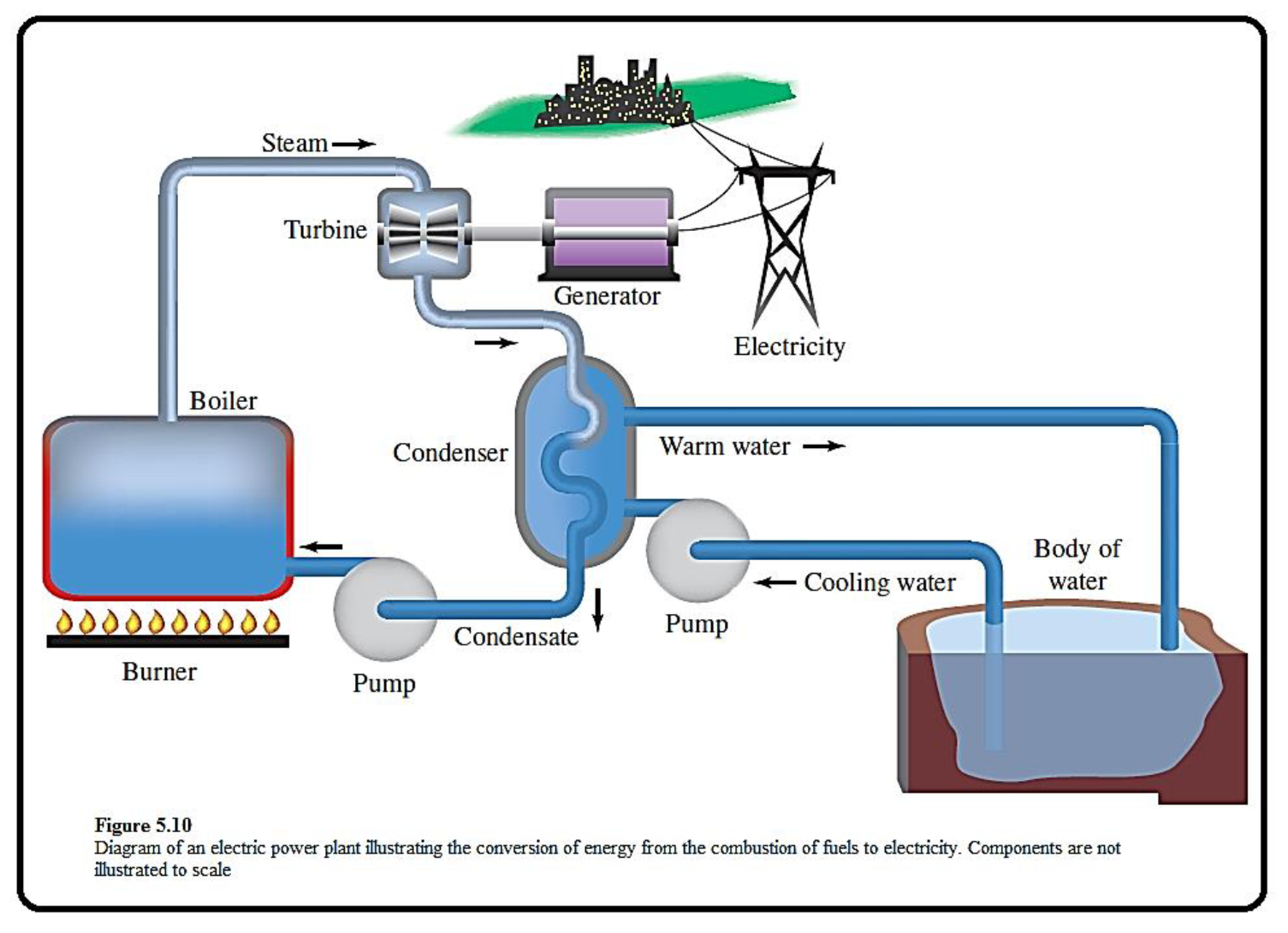
Figure 1
Coal is a best fuel to produce heat energy, the steam produced through this process is used to generate electricity.
Energy exists in different forms in our world.
In the given representation, the fuel in the burner is a source of potential energy. When it burned, some of its potential energy is converted to heat throgh combustion. This heat is converted into kinetic energy of steams. By spinning the turbine, the kinetic energy of the steam is converted to mechanical energy. The mechanical energy generated from the spinning turbne is converted to electrical energy by rotating a wire in a magnetic field. This electrical energy, carried to the city by the power lines, light bulbs and heats homes.
Want to see more full solutions like this?
Chapter 5 Solutions
Chemistry In Context
Additional Science Textbook Solutions
General, Organic, & Biological Chemistry
Principles of General, Organic, Biological Chemistry
Living by Chemistry
Organic Chemistry (8th Edition)
Chemistry: Atoms First
Thermodynamics, Statistical Thermodynamics, & Kinetics
- xplain why aluminum cans make good storage containers for soft drinks. Styrofoam cups can be used to keep coffee hot and cola cold. How can this be?arrow_forwardAnalyze both of the images in Figure 15.27 in terms ofpotential energy of position, chemical potential energy,kinetic energy, and heat.arrow_forwardWhat quantity of heat energy would have to be applied to a 25.1 -g block of iron in order to raise the temperature of the iron sample by 17.5 °C? (See Table 10.1.)arrow_forward
- What if energy was not conserved? How would this affect our lives?arrow_forwardEnthalpy a A 100.-g sample of water is placed in an insulated container and allowed to come to room temperature at 21C. To heat the water sample to 41C, how much heat must you add to it? b Consider the hypothetical reaction,2X(aq)+Y(l)X2Y(aq)being run in an insulated container that contains 100. g of solution. If the temperature of the solution changes from 21C to 31C, how much heat does the chemical reaction produce? How does this answer compare with that in part a? (You can assume that this solution is so dilute that it has the same heat capacity as pure water.) c If you wanted the temperature of 100. g of this solution to increase from 21C to 51C, how much heat would you have to add to it? (Try to answer this question without using a formula.) d If you had added 0.02 mol of X and 0.01 mol of Y to form the solution in part b, how many moles of X and Y would you need to bring about the temperature change described in part c. e Judging on the basis of your answers so far, what is the enthalpy of the reaction 2X(aq) + Y(l) X2Y(aq)?arrow_forwardHydrogen chloride gas dissolves in water to form hydrochloric acid (an ionic solution). HCl(g)H2OH+(aq)+Cl(aq) Find H for the above reaction. The data are given in Table 6.2.arrow_forward
- Consider the reaction B2H6(g)+3O2(s)B2O3(s)+3H2O(g)H=2035KJ Calculate the amount of heat released when 54.0 g of diborane is combusted.arrow_forwardConsider the following diagram when answering the questions below. a. Compare balls A and B in terms of potential energy in both the initial and final setups. b. Ball A has stopped moving in the figure on the right above, but energy must be conserved. What happened to the potential energy of ball A?arrow_forwardFor the reaction HgO(s)Hg(l)+12O2(g),H=+90.7KJ: a. What quantity of heat is required to produce 1 mole of mercury by this reaction? b. What quantity of heat is required to produce 1 mole of oxygen gas by this reaction? c. What quantity of heat would be released in the following reaction as written? 2Hg(l)+O2(g)2HgO(s)arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





