
Interpretation:
Ozone absorbs UV radiation of wavelengths less than 320 nm, while oxygen requires higher – energy
Concept Introduction:
Electromagnetic spectrum is a range of all types of electromagnetic radiation.
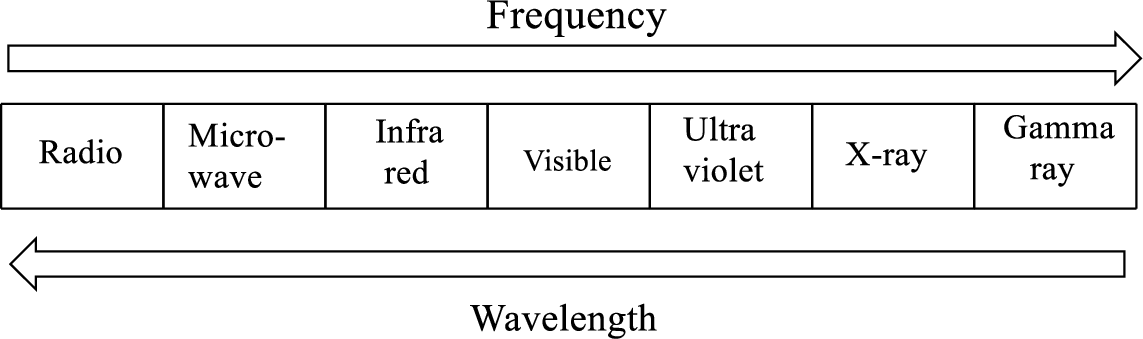
Radiant energy emitted by all matter whose temperature is greater than absolute zero
(
Radiant energy with short wavelength (higher frequency) will have the most energy and as the wavelength get longer, the energy decreases.
Bond energy or more correctly the bond dissociation enthalpy is the enthalpy change when breaking a bond in a molecule with the reactant and products in the gas phase.
Resonance structures: when a single Lewis structure does not adequately represent a substance, the true structure is intermediate between two or more structures which are called resonance structures and these structures are created by moving electrons, not atoms.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Chemistry In Context
- A Lewis structure obeying the octet rule can be drawn for O2 as follows: Use the molecular orbital energy-level diagram for O2 to show that the above Lewis structure corresponds to an excited state.arrow_forwardDraw all of the Lewis structures of O22- which obey the octet rule and use this Lewis structure or these resonance structures to predict how many covalent bonds connect each oxygen atom in the real structure to the central O atom. Assume that the octet rule is followed for the O atom when you draw your structure(s). Pick the correct statement from the choices below. a) Each oxygen atom is connected to the central O atom with 1 covalent bonds. b) Each oxygen atom is connected to the central O atom with 1.33 covalent bonds. c) Each oxygen atom is connected to the central O atom with 2 covalent bonds. d) Each oxygen atom is connected to the central O atom with 1.67 covalent bonds. e) Each oxygen atom is connected to the central O atom with 1.25 covalent bonds.arrow_forwardUsing bond energies, explain the reaction sequence of ozone-destroying capability in the stratosphere of hydrocarbons containing the halogens: Br > Cl> Farrow_forward
- A nonmetal like oxygen forms both ionic and covalent bonds, depending on the identity of the element to which it bonds. What type of bonding is observed in CaO and CO 2? Explain why two different types of bonding are observed.arrow_forward18 19 20 21 22 23 24 Answer the questions in the table below about the shape of the methyl (CH3) anion. How many electron groups are around the central carbon atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. 0 What phrase best describes the arrangement of these electron groups around the central carbon atom? (You may need to use the scrollbar to see all the choices.) ✓ (choose one) linear bent T-shaped trigonal planar trigonal pyramidal square planar square pyramidal tetrahedral sawhorse trigonal bipyramidal octahedral Continue Two Column N....pages Kindergarten O....docx Grey Black and B....pdf 14 431 10 25 nola Mir Grey Black and B....pdf 26 Ⓒ2022 McGraw Hill LLC. Allarrow_forwardGalium (Ga) is an element in group III with atomic number 31. By referring to the Periodic Table, answer the following questions. Explain how the atomic radius of gallium differs from aluminium. Explain how the electronegativity of gallium differs from aluminium. Explain how the electronegativity of gallium differs from germanium. Explain how the ionisation energy of gallium differs from germanium. Explain how the ionisation energy of gallium differs from indium.arrow_forward
- Silicon is in the same group of the periodic table as carbon and,like carbon, can form up to four single bonds. Many science fiction stories have been based on the premise of silicon-based life. Is this realistic? What characteristics of silicon make it less well adapted than carbon as the central organizing element for life? To answer this question, consider what you have learned about carbon’s bonding versatility, and refer to a beginning inorganic chemistry textbook for silicon’s bonding propertiesarrow_forwardDraw Lewis structures of the peroxide ion and the oxygen moleculearrow_forwardHydrazine, N2H4, burns in oxygen as follows: N2H4 + O2 → N2 + 2H2O [The bond energies in kJ/mol are: N-H = 388; N-N 163; N≡N 944; O-H 463; O=O 496] Draw the chemical structures of the reactants and products and give the formula to calculate enthalpy change in a reaction, ΔH.arrow_forward
- The pentafluorides of the larger members of Group 5A(15)have been prepared, but N can have only eight electrons. A claim has been made that, at low temperatures, a compound with the empirical formula NF₅ forms. Draw a possible Lewis structure for this compound.arrow_forwardWould a photon whose wavelength is 145 nm have enough energy to photodissociate O2 whose bond energy is 495 kJ/mol? Would it have enough energy to photoionize O2?arrow_forwardAlthough PF5 and AsF5 are stable, nitrogen does not form NF5 molecules. Explain this difference among members of the same group.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning




