
Organic Chemistry
4th Edition
ISBN: 9780073402772
Author: Janice G. Smith
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 5.58P
Explain each statement by referring to compounds A-E.
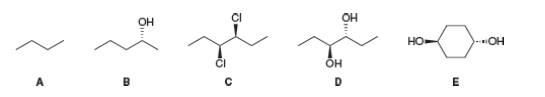
a. A has a mirror image bur no enantiomer.
b. B has an enantiomer and no diastereomer.
c. C has both an enantiomer and a diastereomer.
d. D has a diastereomer but no enantiomer.
e. E has a diastereomer but no enantiomer.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain each statement by referring to compounds A-E.
он
он
HO
.OH
CI
OH
A
в
E
a. A has a mirror image but no enantiomer.
b. B has an enantiomer and no diastereomer.
c. C has both an enantiomer and a diastereomer.
d. D has a diastereomer but no enatiomer.
e. E has a diastereomer but no enantiomer.
E..
1. An object is chiral if it
A. is superimposable on its mirror image
B. is not superimposable on its mirror image
C. has no mirror image
D. is identical to each mirror image of itself
Match the pair of compounds with the type of isomerismChoices:
a. Functional Isomer
b. Positional isomer
c. Enantiomer
d. E,Z configuration
e. Skeletal Isomer
f. cis, trans configuration
g. diastereomer
Chapter 5 Solutions
Organic Chemistry
Ch. 5 - Prob. 5.1PCh. 5 - Prob. 5.2PCh. 5 - Draw the mirror image of each compound. Label each...Ch. 5 - Prob. 5.4PCh. 5 - Prob. 5.5PCh. 5 - Prob. 5.6PCh. 5 - The facts in section 5.4A can be used to locate...Ch. 5 - Prob. 5.8PCh. 5 - Prob. 5.9PCh. 5 - Prob. 5.10P
Ch. 5 - Prob. 5.11PCh. 5 - Prob. 5.12PCh. 5 - Label each compound as R or S.Ch. 5 - Prob. 5.14PCh. 5 - Prob. 5.15PCh. 5 - Prob. 5.16PCh. 5 - Prob. 5.17PCh. 5 - Prob. 5.18PCh. 5 - Prob. 5.19PCh. 5 - Prob. 5.20PCh. 5 - Prob. 5.21PCh. 5 - Prob. 5.22PCh. 5 - Prob. 5.23PCh. 5 - Which of the following cyclic molecules are meso...Ch. 5 - Prob. 5.25PCh. 5 - Prob. 5.26PCh. 5 - The amino acid (S)-alanine has the physical...Ch. 5 - Prob. 5.28PCh. 5 - Prob. 5.29PCh. 5 - Prob. 5.30PCh. 5 - Prob. 5.31PCh. 5 - Prob. 5.32PCh. 5 - Prob. 5.33PCh. 5 - Prob. 5.34PCh. 5 - Prob. 5.35PCh. 5 - Prob. 5.36PCh. 5 - Prob. 5.37PCh. 5 - Determine if each compound is identical to or an...Ch. 5 - Prob. 5.39PCh. 5 - Locate the stereogenic centers in each compound. A...Ch. 5 - Prob. 5.52PCh. 5 - Prob. 5.41PCh. 5 - Prob. 5.42PCh. 5 - Prob. 5.43PCh. 5 - Prob. 5.44PCh. 5 - Prob. 5.45PCh. 5 - Label each stereogenic center as R or S. a. c. e....Ch. 5 - Prob. 5.47PCh. 5 - Prob. 5.48PCh. 5 - Prob. 5.49PCh. 5 - Prob. 5.50PCh. 5 - Prob. 5.51PCh. 5 - Prob. 5.53PCh. 5 - Prob. 5.54PCh. 5 - Prob. 5.55PCh. 5 - Draw all possible stereoisomers for each...Ch. 5 - Prob. 5.57PCh. 5 - 5.59 Explain each statement by referring to...Ch. 5 - Prob. 5.59PCh. 5 - Prob. 5.60PCh. 5 - Prob. 5.61PCh. 5 - Prob. 5.62PCh. 5 - Prob. 5.63PCh. 5 - Prob. 5.64PCh. 5 - Prob. 5.65PCh. 5 -
5.67 Artemisinin and mefloquine are widely used...Ch. 5 - 5.68 Saquinavir (trade name Invirase) is a...Ch. 5 - Prob. 5.68PCh. 5 - Prob. 5.69PCh. 5 - Prob. 5.70PCh. 5 - Prob. 5.71PCh. 5 - Problem 5.73 An acid-base reaction of with a...
Additional Science Textbook Solutions
Find more solutions based on key concepts
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living by Chemistry
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 8) Identify enantiomers. C E A CI H ICL F CI H Cle H. CI H. CI H CI CI H. F H. 'F 'F H. F H H. F H H H. A) B and D, A and C B) B and D C) A and B, A and D, B and C, C and D D) B and D, A and B, A and D, B and C, C and D E) A and Carrow_forwardThese two products are a. diastereomers b. constitutional isomers c. identicalarrow_forwardMatch the pairs of compounds with the type of isomerism a. functional isomer b. skeletal isomer c. positional isomer d. cis, trans configuration e. diastereomer f. e,z, configuration g. enantiomerarrow_forward
- Saquinavir (trade name Invirase) is a protease inhibitor, used to treat HIV (human immunodeficiency virus). a.Locate all stereogenic centers in saquinavir, and label each stereogenic center as R or S. b.Draw the enantiomer of saquinavir. c.Draw a diastereomer of saquinavir. d.Draw a constitutional isomer that contains at least one different functional group.arrow_forwardShow two different rearrangement products for the carbocation shown below. Circle the rearrangement product that is preferred (more likely to form). 04 Attach File Browse Local Filesarrow_forward1. Identify the relationship between the following two structures.a. Identicalb. neitherc. diastereomers2. Identify the relationship between the following two structures.a. enantiomersb. diastereomersarrow_forward
- 5. CIRCLE ALL THAT APPLY: Why is it so difficult to physically separate enantiomers from each other in a mixture? a. They are the same compound b. They have the same melting points c. They have the same densities d. They are both meso compoundsarrow_forwardA. Draw the enantiomer of the following compound. B. Draw a diastereomer of the following compound. „CI H. H2N"arrow_forward17. Enantiomers of CHBrIF Perspective drawings of both enantiomers: a. Make models of both enantiomers (make one model then make its mirror image.) b. Can the two models be superimposed? c. What type of isomerism do these models represent? 18. Geometric Isomers of CICH = CHCI (1,2-dichloroethene) Perspective drawings of both isomers: a. Make two models that have different spatial arrangement of the substitutions (the two Cl atoms) across the double bond. b. Draw the perspective formulae for the two isomers and label them with their correct designations. c. What type of isomerism do these represent?arrow_forward
- What is the significance of a reaction that occurs in 94% enantiomeric excess? A. The product contains 94% of one enantiomer and 6% of other products B. The product contains 94% of one enantiomer and 6% of another enantiomer C. The product contains an enantiomer that is 94% pure. D. Product contains 97% of one enantiomer and 3% of another enantiomerarrow_forwardLocate the stereogenic center in each compound and draw both enantiomers. b. HO, OH С. HO. NH2 a.arrow_forwardDrawn are four isomeric dimethylcyclopropanes. a.How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D? b.Label each compound as chiral or achiral. c.Which compounds alone would be optically active? d.Which compounds have a plane of symmetry? e.How do the boiling points of the compounds in each pair compare: A and B; B and C; C and D? f.Which of the compounds are meso compounds? g.Would an equal mixture of compounds C and D be optically active? What about an equal mixture of B and C?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License