
Concept explainers
(a)
Interpretation:
Missing curved arrows are to be supplied for the given proton transfer reaction. The relevant electrons are to be drawn if they are not shown.
Concept introduction:
In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid to a Bronsted-Lowry base in a single elementary step in which one bond is broken and another is formed simultaneously. The curved arrow notation shows the movement of valence electrons, not atoms. The movement of two electrons is shown be using a double-barbed arrow. To represent bond breaking, the tail of the arrow originates from the center of a bond whereas to represent bond formation, the head of arrow points to an atom which forms the new bond, that is, σ bond or the region where the bond is formed if the new bond is a π bond.
Answer to Problem 6.39P
The missing curved arrow notation for the proton transfer reaction and relevant electrons is shown as
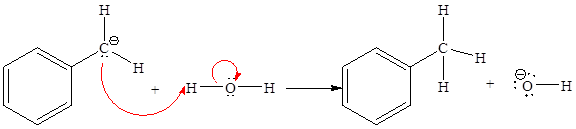
Explanation of Solution
The given proton transfer reaction is
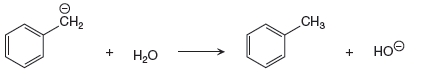
In the above reaction, the bond
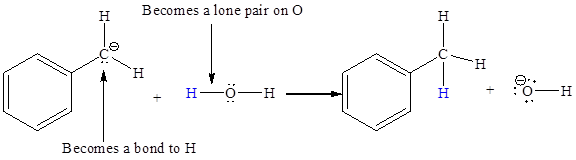
The appropriate movement of these valence electrons is shown by using curved arrow notations. One curved arrow is to be drawn from the lone pair on C to the H on water (highlighted blue) to illustrate the formation of
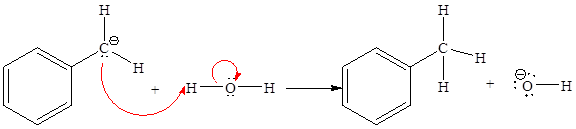
The curved arrow notation for the proton transfer of the given reaction is drawn on the basis of the movement of valence electrons involved in bond breaking and bond formation.
(b)
Interpretation:
Missing curved arrows are to be supplied for the given proton transfer reaction. The relevant electrons are to be drawn if they are not shown.
Concept introduction:
In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid to a Bronsted-Lowry base in a single elementary step in which one bond is broken and another is formed simultaneously. The curved arrow notation shows the movement of valence electrons, not atoms. The movement of two electrons is shown be using a double-barbed arrow. To represent bond breaking, the tail of the arrow originates from the center of a bond whereas to represent bond formation, the head of arrow points to an atom which forms the new bond, that is, ![]() bond or the region where the bond is formed if the new bond is a
bond or the region where the bond is formed if the new bond is a ![]() bond.
bond.
Answer to Problem 6.39P
The missing curved arrow notation for the proton transfer reaction and relevant electrons is shown as
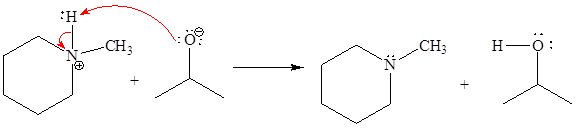
Explanation of Solution
The given proton transfer reaction is
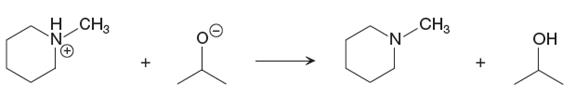
In the above reaction, the bond
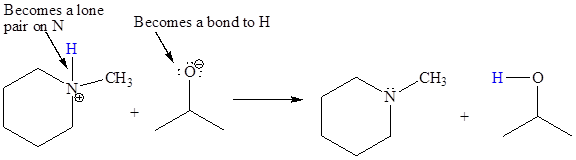
The appropriate movement of these valence electrons is shown by using curved arrow notations. One curved arrow is to be drawn from the lone pair on O to the H on N (highlighted blue) to illustrate the formation of the

The curved arrow notation for the proton transfer of the given reaction is drawn on the basis of the movement of valence electrons involved in bond breaking and bond formation.
(c)
Interpretation:
Missing curved arrows are to be supplied for the given proton transfer reaction. The relevant electrons are to be drawn if they are not shown.
Concept introduction:
In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid to a Bronsted-Lowry base in a single elementary step in which one bond is broken and another is formed simultaneously. The curved arrow notation shows the movement of valence electrons, not atoms. The movement of two electrons is shown be using a double-barbed arrow. To represent bond breaking, the tail of the arrow originates from the center of a bond whereas to represent bond formation, the head of arrow points to an atom which forms the new bond, that is, ![]() bond or the region where the bond is formed if the new bond is a
bond or the region where the bond is formed if the new bond is a ![]() bond.
bond.
Answer to Problem 6.39P
The missing curved arrow notation for the proton transfer reaction and relevant electrons is shown as
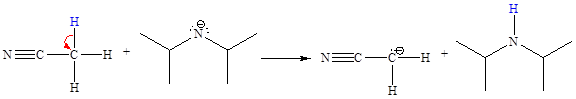
Explanation of Solution
The given proton transfer reaction is
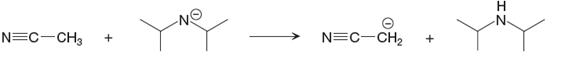
In the above reaction, the bond
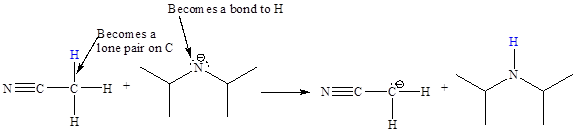
The appropriate movement of these valence electrons is shown by using curved arrow notations. One curved arrow is to be drawn from the lone pair on N to the H on C (highlighted blue) to illustrate the formation of the
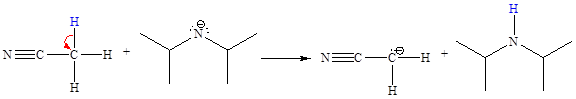
The curved arrow notation for the proton transfer of the given reaction is drawn on the basis of the movement of valence electrons involved in bond breaking and bond formation.
(d)
Interpretation:
Missing curved arrows are to be supplied for the given proton transfer reaction. The relevant electrons are to be drawn if they are not shown.
Concept introduction:
In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid to a Bronsted-Lowry base in a single elementary step in which one bond is broken and another is formed simultaneously. The curved arrow notation shows the movement of valence electrons, not atoms. The movement of two electrons is shown be using a double-barbed arrow. To represent bond breaking, the tail of the arrow originates from the center of a bond whereas to represent bond formation, the head of arrow points to an atom which forms the new bond, that is, ![]() bond or the region where the bond is formed if the new bond is a
bond or the region where the bond is formed if the new bond is a ![]() bond.
bond.
Answer to Problem 6.39P
The missing curved arrow notation for the proton transfer reaction and relevant electrons is shown as
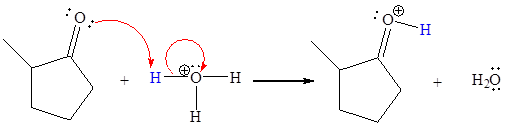
Explanation of Solution
The given proton transfer reaction is
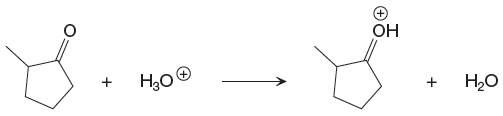
In the above reaction, the bond
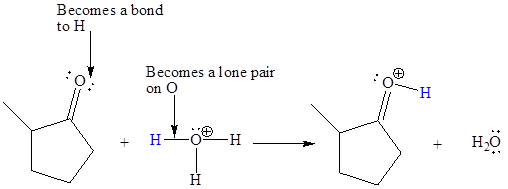
The appropriate movement of these valence electrons is shown by using curved arrow notations. One curved arrow is to be drawn from the lone pair on O to the H of
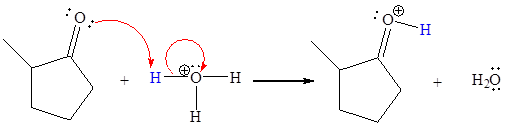
The curved arrow notation for the proton transfer of the given reaction is drawn on the basis of the movement of valence electrons involved in bond breaking and bond formation.
Want to see more full solutions like this?
Chapter 6 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- a) Draw the mechanism and products of the acid-base reaction shown below. b) Identify the role of each reactant and product: acid, base, conjugate acid, conjugate base andclearly indicate using the reversible unequal arrows whether the equilibrium favors the reactants or the products c) Justify your answer indicated in (c) using the concepts seen in class. One argument is sufficientarrow_forwardcan you explain in paragraph, how the current reaction is taking place, and each step.arrow_forwardDraw the product of the Lewis acid-base reaction shown in the picture attached and include formal charges/lone pairs. Draw any curly arrows to show electron flowarrow_forward
- Need both sets of compounds ranked strongest to weakestarrow_forwardI understand where the proton transfer occurs but not what the products will look like. Can you draw curved arrows to show proton transfer and what the reaction products would look like? And explain which side is favored and whyarrow_forwardGive typing answer with explanation and conclusion Which from the list below can affect the rate of a reaction? (Choose all that apply). Group of answer choices heat light buffers enzymes temperaturearrow_forward
- Identify all the ACIDS in this sequence of reactions (both fwd and reverse directions) (Can be multiple answers)arrow_forwardlight in red each acidic location on the organic molecule at left. Highlight in blue each basic location on the organic molecule at r e for advanced students: we mean acidic or basic in the Brønsted-Lowry sense only. HO OH HO INI OH èarrow_forwardHi, For this proton transfer, why does the NH3 electron pair move to the H and not the electron pair from the Br?arrow_forward
- Which is the least and which is the most reactive for hydrolysisarrow_forwardCircle the side that favored at equilibrium for the acid base reactions.arrow_forward(Intermediate) Reactant (Intermediate) Product H3C 'o: Apply Mechanism Hint Solution 2 remaining step(s) can be solved Tip: Only add curved arrows in this sketcherarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning


