
Concept explainers
(a)
Interpretation:
The most acidic proton, in the given species, is to be identified, and its pKa value is to be estimated.
Concept introduction:
An Acidic proton is one which is directly bonded to an electronegative atom. The acidity of a compound is governed largely by the
Answer to Problem 6.50P
The most acidic proton in the given species along with its estimated pKa value is:
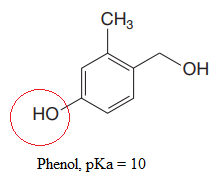
Explanation of Solution
The structure for the given compound is:
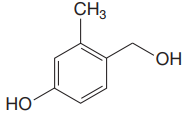
There are three protons that could be acidic. The proton attached to the carbon in methyl group, to the oxygen atom in alcohol, and the proton directly attached to the oxygen atom in phenol functional group are the protons that could be acidic.
According to Table 6-1, the relative pKa value of each of the protons is:
The pKa value for the compound having
The pKa value for the compound having
The pKa value for the compound having
Lower the pKa value, stronger is the acid, and the proton associated with it is the most acidic proton. The lowest pKa value is for

The most acidic proton in the given structure is identified along with its estimated pKa value using Table 6-1.
(b)
Interpretation:
The most acidic proton in the given species is to be identified and its pKa value is to be estimated.
Concept introduction:
An Acidic proton is the one which is directly bonded to an electronegative atom. The acidity of a compound is governed largely by the functional group on which the acidic proton is found. Nearby structural features such as highly electronegative substituent or presence of a double or triple bond can alter the acidity significantly. The pKa value for a particular compound is explained based on structural similarities of the compound and the compounds listed in Table 6-1.
Answer to Problem 6.50P
The most acidic proton in the given species along with its estimated pKa value is:
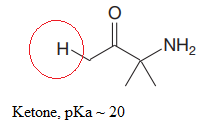
Explanation of Solution
The structure for the given compound is:
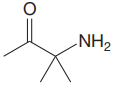
In the given structure, the proton attached to the nitrogen atom, and to the carbon atom next to the carbonyl group, could be the acidic protons.
According to Table 6-1, the relative pKa value of each of the protons is:
The pKa value for the compound having
The pKa value for the compound having
Lower the pKa value, stronger is the acid, and the proton associated with it is the most acidic proton. The lowest pKa value is for

The most acidic proton in the given structure is identified along with its estimated pKa value using Table 6-1.
(c)
Interpretation:
The most acidic proton in the given species is to be identified and its pKa value is to be estimated.
Concept introduction:
An Acidic proton is the one which is directly bonded to an electronegative atom. The acidity of a compound is governed largely by the functional group on which the acidic proton is found. Nearby structural features such as highly electronegative substituent or presence of a double or triple bond can alter the acidity significantly. The pKa value for a particular compound is explained based on structural similarities of the compound and the compounds listed in Table 6-1.
Answer to Problem 6.50P
The most acidic proton in the given species along with its estimated pKa value is:
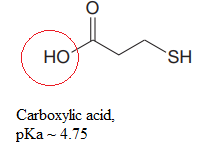
Explanation of Solution
The structure for the given compound is:
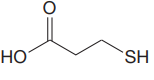
There are two protons that could be acidic. The proton attached to the oxygen atom which is directly bonded to the carbonyl group, and to the sulfur atom are the protons that could be acidic.
According to Table 6-1, the relative pKa value of each of the protons is:
The pKa value for the compound having
The pKa value for the compound having
Lower the pKa value, stronger is the acid and the proton associated with it is the most acidic proton. The lowest pKa value is for

The most acidic proton in the given structure is identified along with its estimated pKa value using Table 6-1.
(d)
Interpretation:
The most acidic proton in the given species is to be identified and its pKa value is to be estimated.
Concept introduction:
An Acidic proton is the one which is directly bonded to an electronegative atom. The acidity of a compound is governed largely by the functional group on which the acidic proton is found. Nearby structural features such as highly electronegative substituent or presence of a double or triple bond can alter the acidity significantly. The pKa value for a particular compound is explained based on structural similarities of the compound and the compounds listed in Table 6-1.
Answer to Problem 6.50P
The most acidic proton in the given species along with its estimated pKa value is:
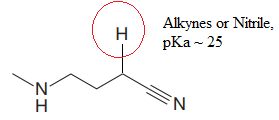
Explanation of Solution
The structure for the given compound is:
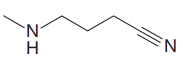
In the given structure, the proton attached to the nitrogen atom and to the triple bonded carbon atom could be the acidic protons.
According to Table 6-1, the relative pKa value of each of the protons is:
The pKa value for the compound having
The pKa value for the compound having
Lower the pKa value, stronger is the acid and the proton associated with it is the most acidic proton. The lowest pKa value is for

The most acidic proton in the given structure is identified along with its estimated pKa value using Table 6-1.
(e)
Interpretation:
The most acidic proton in the given species is to be identified and its pKa value is to be estimated.
Concept introduction:
An Acidic proton is the one which is directly bonded to an electronegative atom. The acidity of a compound is governed largely by the functional group on which the acidic proton is found. Nearby structural features such as highly electronegative substituent or presence of a double or triple bond can alter the acidity significantly. The pKa value for a particular compound is explained based on structural similarities of the compound and the compounds listed in Table 6-1.
Answer to Problem 6.50P
The most acidic proton in the given species along with its estimated pKa value is:
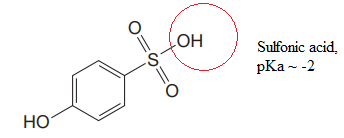
Explanation of Solution
The structure for the given compound is:
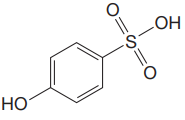
There are two protons that could be acidic. The proton attached to the carbon in methyl group, to the oxygen atom in alcohol, and the proton directly attached to the oxygen atom in phenol functional group are the protons that could be acidic.
According to Table 6-1, the relative pKa value of each of the protons is:
The pKa value for the compound having
The pKa value for the compound having
Lower the pKa value, stronger is the acid and the proton associated with it is the most acidic proton. The lowest pKa value is for

The most acidic proton in the given structure is identified along with its estimated pKa value using Table 6-1.
(f)
Interpretation:
The most acidic proton in the given species is to be identified and its pKa value is to be estimated.
Concept introduction:
An Acidic proton is the one which is directly bonded to an electronegative atom. The acidity of a compound is governed largely by the functional group on which the acidic proton is found. Nearby structural features such as highly electronegative substituent or presence of a double or triple bond can alter the acidity significantly. The pKa value for a particular compound is explained based on structural similarities of the compound and the compounds listed in Table 6-1.
Answer to Problem 6.50P
The most acidic proton in the given species along with its estimated pKa value is:
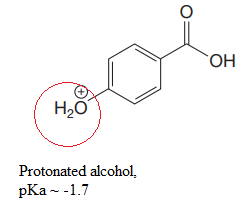
Explanation of Solution
The structure for the given compound is:
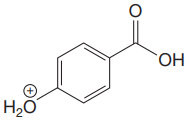
There are two protons that could be acidic. The proton attached to the carbon in methyl group, to the oxygen atom in alcohol, and the proton directly attached to the oxygen atom in phenol functional group are the protons that could be acidic.
According to Table 6-1, the relative pKa value of each of the protons is:
The pKa value for the compound having protonated
The pKa value for the compound having
Lower the pKa value, stronger is the acid and the proton associated is the most acidic proton. The lowest pKa value is for protonated

The most acidic proton in the given structure is identified along with its estimated pKa value using Table 6-1.
(g)
Interpretation:
The most acidic proton in the given species is to be identified and its pKa value is to be estimated.
Concept introduction:
An Acidic proton is the one which is directly bonded to an electronegative atom. The acidity of a compound is governed largely by the functional group on which the acidic proton is found. Nearby structural features such as highly electronegative substituent or presence of a double or triple bond can alter the acidity significantly. The pKa value for a particular compound is explained based on structural similarities of the compound and the compounds listed in Table 6-1.
Answer to Problem 6.50P
The most acidic proton in the given species along with its estimated pKa value is:
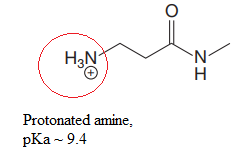
Explanation of Solution
The structure for the given compound is:
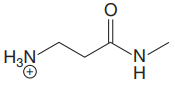
In the given structure, the proton attached to both nitrogen atoms could be acidic protons.
According to Table 6-1, the relative pKa value of each of the protons is:
The pKa value for the compound having protonated
The pKa value for the compound having
Lower the pKa value, stronger is the acid and the proton associated with it is the most acidic proton. The lowest pKa value is for protonated

The most acidic proton in the given structure is identified along with its estimated pKa value using Table 6-1.
(h)
Interpretation:
The most acidic proton in the given species is to be identified and its pKa value is to be estimated.
Concept introduction:
An Acidic proton is the one which is directly bonded to an electronegative atom. The acidity of a compound is governed largely by the functional group on which the acidic proton is found. Nearby structural features such as highly electronegative substituent or presence of a double or triple bond can alter the acidity significantly. The pKa value for a particular compound is explained based on structural similarities of the compound and the compounds listed in Table 6-1.
Answer to Problem 6.50P
The most acidic proton in the given species along with its estimated pKa value is:
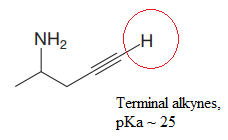
Explanation of Solution
The structure for the given compound is:
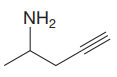
In the given structure, the protons attached to the terminal triple bonded carbon atom and to the nitrogen atom could be acidic protons.
According to Table 6-1, the relative pKa value of each of the protons is:
The pKa value for the compound having protonated
The pKa value for the compound having
Lower the pKa value, stronger is the acid and the proton associated with it is the most acidic proton. The lowest pKa value is for

The most acidic proton in the given structure is identified along with its estimated pKa value using Table 6-1.
Want to see more full solutions like this?
Chapter 6 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Identify which of the following compounds is expected to be a stronger base. Justify your choicearrow_forwardRank the following conjugate bases in order of decreasing basicity, putting the most basic first. H2C=CH HC=C CH3 II Multiple Choice || > |> |II |> || > III III > || > | III > | > ||arrow_forward(a) Given that Ka for acetic acid is 1.8 x 10-5 and that forhypochlorous acid is 3.0 x 10-8, which is the stronger acid?(b) Which is the stronger base, the acetate ion or the hypochloriteion? (c) Calculate Kb values for CH3COO- and ClO-.arrow_forward
- Arrange the numbered protons in decreasing order of acidity, explain the reasoning based upon the conjugate bases.arrow_forwardPlace the following in order of increasing basicityarrow_forwardcomplete the equation for the reaction between each Lewis acid-base pair. In each equation, label which starting material is the Lewis acid and which is the Lewis base; use curved arrows to show the flow of electrons in each reaction. In doing this problem, it is essential that you show valence electrons for all atoms participating in each reactionarrow_forward
- (a) Given that Kb for ammonia is 1.8 x10-5 and that forhydroxylamine is 1.1 x 10-8, which is the stronger base?(b) Which is the stronger acid, the ammonium ion or thehydroxylammonium ion? (c) Calculate Ka values for NH4+and H3NOH+.arrow_forwardWhich compound would have the least stable conjugate base and why?arrow_forwardRank the following compounds in terms of increasing acidity (least acidic first). Explain your ranking. Making sure to say which hydrogen in each molecule is the most acidic, and discuss the relative stability of the conjugate bases.arrow_forward
- why are the values of E on the two bases related to each other in such a simple wayarrow_forwardPut the following bases in order from weakest to strongest and explain why. Cute specific pKa values of conjugate acids to answer this question.arrow_forwardWhich of the following compounds would be the strongest base?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
