
Concept explainers
(a)
Interpretation:
The complete, detailed mechanism and products for the given reaction are to be drawn assuming that it takes place via an
Concept introduction:
Answer to Problem 8.13P
The complete, detailed mechanism for the given reaction, assuming that it takes place via an
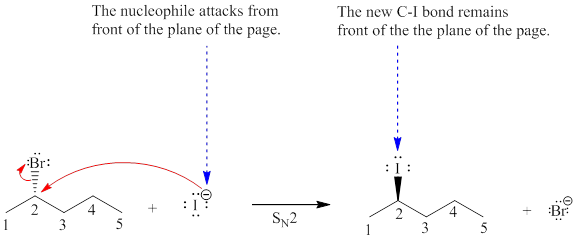
Explanation of Solution
The given reaction is:
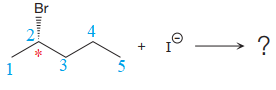
The reactant molecule contains one chiral center at the C2 carbon atom. The leaving group is

In given alkyl halide C2 carbon atom is chiral in nature and affected throughout the course of the reaction. The nucleophile
For the given
(b)
Interpretation:
The complete, detailed mechanism and products for the given reaction are to be drawn assuming that it takes place via an
Concept introduction:
Answer to Problem 8.13P
The complete, detailed mechanism for the given reaction, assuming that it takes place via an
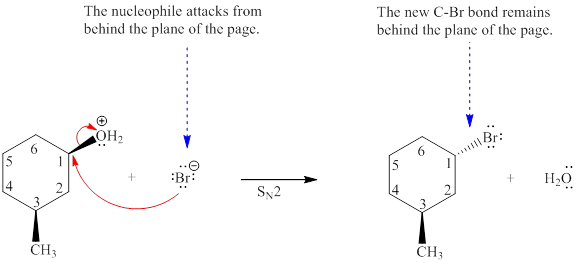
Explanation of Solution
The given reaction is
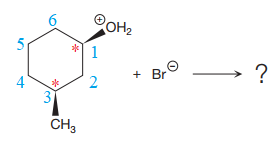
The reactant molecule contains two chiral centers at C1 and C3 carbon atoms. The leaving group is

Note that the stereochemical configuration at the chiral center at C3 remains unchanged because no bonds to it were broken or formed. However, the stereochemical configuration at the chiral center at C1 changes as bonds to it are affected throughout the course of reaction. This will change the stereochemical configuration at C3 chiral carbon atom. Thus, a single stereoisomer will be generated.
For the given
(c)
Interpretation:
The complete, detailed mechanism and products for the given reaction are to be drawn assuming that it takes place via an
Concept introduction:
Answer to Problem 8.13P
The complete, detailed mechanism for the given reaction, assuming that it takes place via an
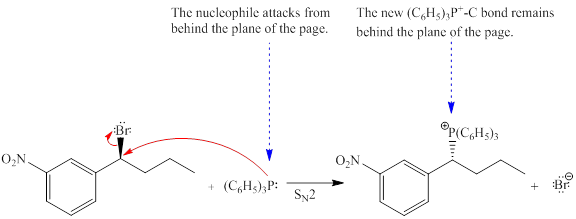
Explanation of Solution
The given reaction is
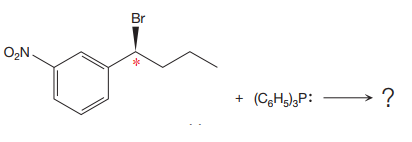
The reactant molecule contains one chiral center. The leaving group is

Note that, the stereochemical configuration at the chiral center changes as bonds to it ae affected throughout the course of reaction. This will change the stereochemical configuration at the chiral center, and it will be opposite than the reactant molecule. Thus, a single stereoisomer will be generated.
For the given
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- For the following reactions draw the full mechanism of each one .arrow_forwardDraw the complete mechanism for the following reaction. Predict the product(s) as well. Br₂arrow_forwardDraw the intermediates A-C for reactions a and b. Please show the appropriate stereochemistry where necessary.arrow_forward
- Draw the major substitution and elimination products for the reaction shown below. You must show the correct stereochemistry where applicable.arrow_forwardDetermine the step by step mechanism of the major product of the following reaction below. Furthermore, determine all-of the product for the following reaction and circle the major product and explain. Clz hvarrow_forwardDraw a detailed mechanism and show the product for the following reaction. HO H₂SO4arrow_forward
- Complete all the reactions/show the products, and for each reaction clearly and thoroughly explain which mechanism (E1, E2, SN1, SN2) is predominant and how it effects the product formation.arrow_forwardConsider the nucleophilic substitution reaction shown here. Based on the stereochemistry, does it proceed by an Sy1 or Sy2 mechanism? Explain. OH KOCH3 Enantiomer CH;OH `OCH3arrow_forwardThe reaction shown here proceeds via a carbocation rearrangement. Draw a complete, detailed mechanism to account for the product. Explain why the carbocation rearrangement is favorable. CH;OH Brarrow_forward
- Please explain the steps from the reactant to product. Are they SN1/SN2/E1 or E2 reactions, good or bad leaving groups.arrow_forward10. Draw the complete, detailed mechanism (curved arrows) for the following reaction, and predict the major product. CH3 ▪OH 1. DMSO (COCI)2 2. Et3N Harrow_forward4. Draw the reaction mechanism and product for the following reaction. Same HO [H*] Quyarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
