
Concept explainers
(a)
Interpretation:
Complete
Concept introduction:
A bimolecular nucleophilic substitution (
Answer to Problem 8.43P
Complete
(i)
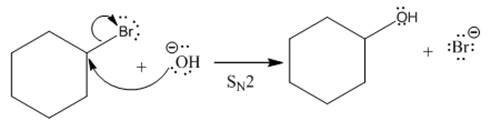
(ii)
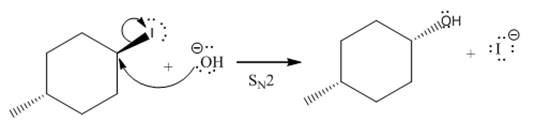
(iii)
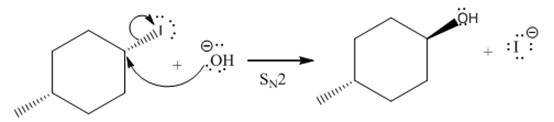
(iv)
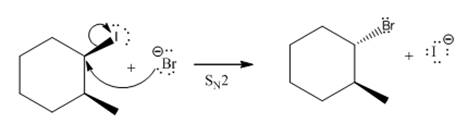
(v)
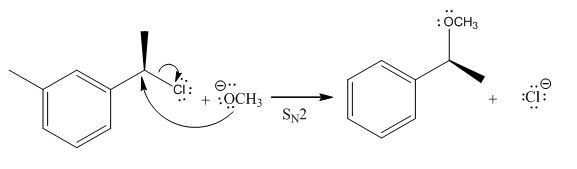
Explanation of Solution
(i)
The given reaction is
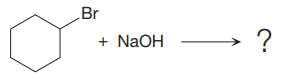
In the above reaction,
(ii)
The given reaction is
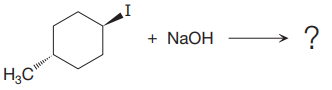
In the above reaction,
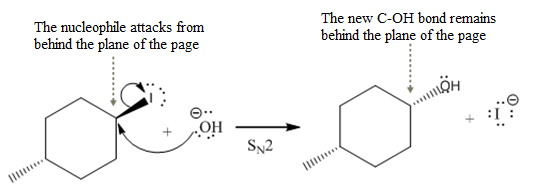
The stereochemistry of another group
(iii)
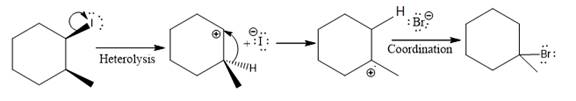
The given reaction
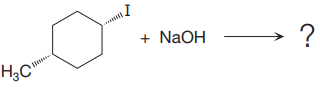
In the above reaction,
The stereochemistry of another group
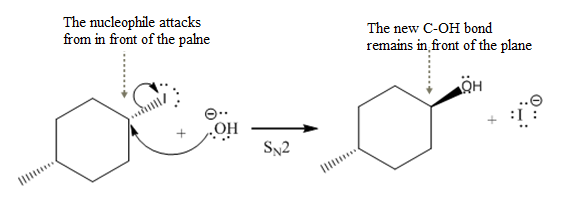
(iv)
The given reaction
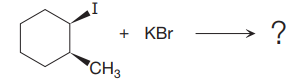
In the above reaction,
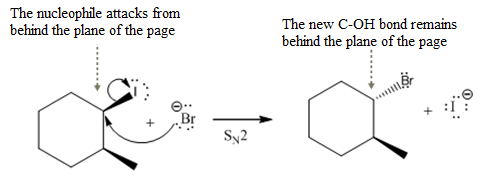
The stereochemistry of another group
(v)
The given reaction
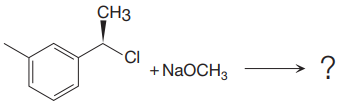
In the above reaction,
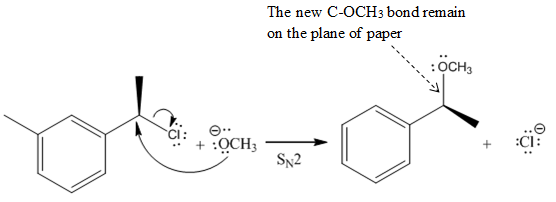
Another group
The Product of
(b)
Interpretation:
Complete
Concept introduction:
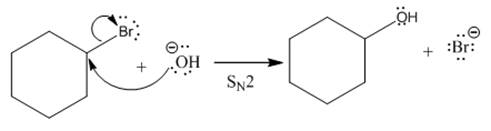
A unimolecular nucleophilic substitution (
If
Answer to Problem 8.43P
Complete
(i)
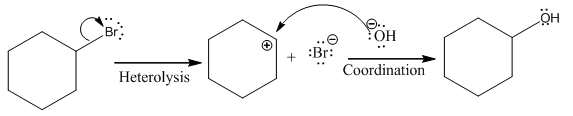
(ii)
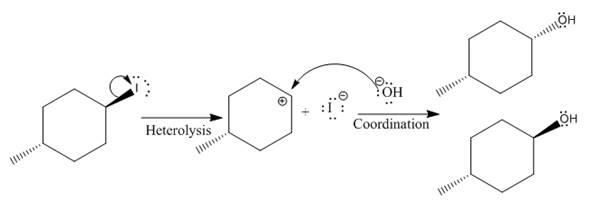
(iii)
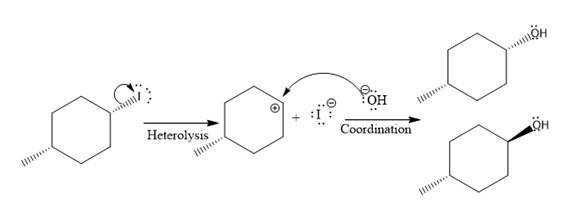
(iv)
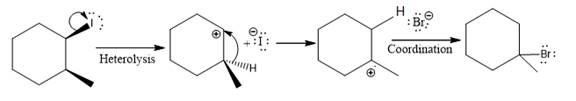
(v)
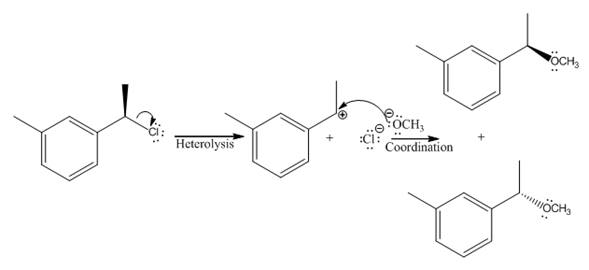
Explanation of Solution
(i)
The given reaction is
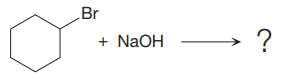

The leaving group in the above reaction is present on the plane of the planar, no stereochemistry is mentioned. Therefore, stereochemistry is not concerned at C, and the only isomer formed is the one shown in the above diagram.
(ii)
The given reaction is
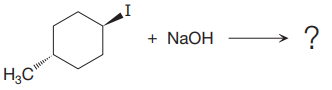
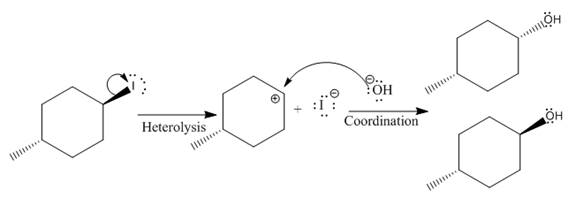
In the above reaction, leaving group (I) is present above the plane of the paper, thus the nucleophile attacks from both sides of the paper- behind the paper and in front of the paper resulting in the formation of two stereoisomers as shown above.
(iii)
The given reaction
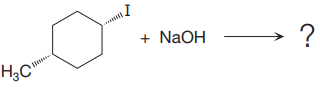

In the above reaction, leaving group (I) is present behind the plane of the paper, thus the nucleophile attacks from both sides of the paper- behind the paper and in front of the paper resulting in the formation of two stereoisomers as shown above.
(iv)
The given reaction
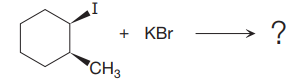
In the above reaction, the carbocation is stabilized by proton transfer. The new carbocation formed at the adjacent carbon from the leaving group is a more stable carbocation. The stereochemistry at the carbon (more stable carbocation) is of no concern. Therefore, the only isomer formed is as shown above.
(v)
The given reaction
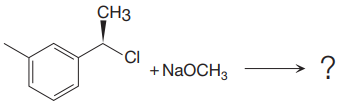
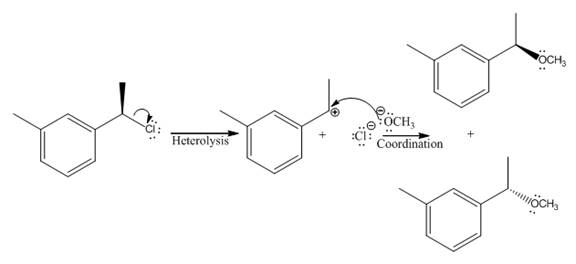
In the above reaction, chiral carbocation is formed once the leaving group leaves. Therefore, the nucleophile can attack from either side- from behind the paper and in front of the paper resulting in the formation of two stereoisomers as shown above.
Product of
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- The reaction shown here is called the pinacol rearrangement. A carbocation rearrangement is believed to be involved. (a) Propose a reasonable mechanism for this reaction. (b) Suggest why the carbocation rearrangement is favorable. HO ОНarrow_forward10. Draw the complete, detailed mechanism (curved arrows) for the following reaction, and predict the major product. CH3 ▪OH 1. DMSO (COCI)2 2. Et3N Harrow_forwardprovide the major product(s) for each of the following reactions. also, provide the mechanism for the last two reactionsarrow_forward
- Consider the intramolecular nucleophilic substitution reaction shown here. Does the stereochemistry of the product suggest an Sn1 or SŅ2 mechanism? Draw the complete mechanism for this reaction, including curved Br OH NaOH CH3 H3C CH3 Br H3C H arrows.arrow_forwardDraw a detailed mechanism for the following addition of HBr across a double bond and explain why the given product is the major product.arrow_forwardProvide the mechanism for each step of the given reaction, showing the major product as well.arrow_forward
- Predict the major thermodynamic and major kinetic product from the following reaction. Label the major products accordingly. Draw the mechanism (arrow pushing) for each product (2 mechanisms total). Please explain stepsarrow_forwardfor each reaction, provide the likely product and the mechanism.arrow_forwardComplete the following reactions by identifying the majority product(s) or the reaction conditions that are missing. No mechanism is neededarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





