
Concept explainers
Write Lewis structures that obey the octet rule (duet rule for H) for each of the following molecules. Carbon is the central atom in CH4, nitrogen is the central atom in NH3, and oxygen is the central atom in H2O.
a. F2
b. O2
c. CO
d. CH4
e. NH3
f. H2O
g. HF
(a)
Interpretation: The Lewis structure is to be drawn for the given molecules.
Concept introduction: The Lewis structure is also known as dot structure. This structure depicts the bonding between atoms and the lone pairs of electrons if exists.
The octet rule states that atoms or molecules gain or lose electrons to get the electronic configuration of nearest noble gas.
To determine: The Lewis structure of the molecule
Answer to Problem 85E
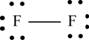
Explanation of Solution
The first step in determining the Lewis structure is to determine the number of valence electrons. The atomic number of fluorine
The valence electron of fluorine is 7
The fluorine molecule
The skeletal structure of
Each fluorine atom requires one electron to complete the octet. Hence, the mutual sharing of two electrons takes place. The 12 valence electrons present are placed as lone pairs in such a way that each atom gets three lone pairs.
The Lewis structure of
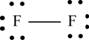
Figure 1
(b)
Interpretation: The Lewis structure is to be drawn for the given molecules.
Concept introduction: The Lewis structure is also known as dot structure. This structure depicts the bonding between atoms and the lone pairs of electrons if exists.
The octet rule states that atoms or molecules gain or lose electrons to get the electronic configuration of nearest noble gas.
To determine: The Lewis structure of the molecule
Answer to Problem 85E
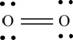
Explanation of Solution
The first step in determining the Lewis structure is to determine the number of valence electrons. The atomic number of oxygen
The valence electron of oxygen is 6
The oxygen molecule
The skeletal structure of
Each oxygen atom requires two electrons to complete the octet. Hence, the mutual sharing of four electrons takes place. The 8 valence electrons present are placed as lone pairs in such a way that each atom gets two lone pairs.
The Lewis structure of
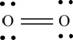
Figure 2
(c)
Interpretation: The Lewis structure is to be drawn for the given molecules.
Concept introduction: The Lewis structure is also known as dot structure. This structure depicts the bonding between atoms and the lone pairs of electrons if exists.
The octet rule states that atoms or molecules gain or lose electrons to get the electronic configuration of nearest noble gas.
To determine: The Lewis structure of the molecule
Answer to Problem 85E

Explanation of Solution
The first step in determining the Lewis structure is to determine the number of valence electrons. The atomic number of oxygen is 8 and its electronic configuration is,
The valence electron of oxygen is 6
The atomic number of carbon
The valence electron of carbon is 4
The molecule
The skeletal structure of
Each oxygen atom requires two electrons to complete the octet whereas carbon requires four electrons to complete the octet. Hence, the mutual sharing of six electrons takes place which is represented by triple bond. The 4 valence electrons left are placed as lone pairs in such a way that each atom gets one lone pair.
The Lewis structure of

Figure 3
(d)
Interpretation: The Lewis structure is to be drawn for the given molecules.
Concept introduction: The Lewis structure is also known as dot structure. This structure depicts the bonding between atoms and the lone pairs of electrons if exists.
The octet rule states that atoms or molecules gain or lose electrons to get the electronic configuration of nearest noble gas.
To determine: The Lewis structure of the molecule
Answer to Problem 85E
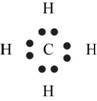
Explanation of Solution
The first step in determining the Lewis structure is to determine the number of valence electrons. The atomic number of carbon is 6 and its electronic configuration is,
The valence electron of carbon is 4
The atomic number of hydrogen
The valence electron of hydrogen is 1
The molecule
The skeletal structure of
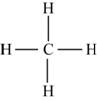
Figure 4
Each hydrogen atom requires one electron to complete the octet whereas carbon requires four electrons to complete the octet. In the molecule
The Lewis structure of

Figure 5
(e)
Interpretation: The Lewis structure is to be drawn for the given molecules.
Concept introduction: The Lewis structure is also known as dot structure. This structure depicts the bonding between atoms and the lone pairs of electrons if exists.
The octet rule states that atoms or molecules gain or lose electrons to get the electronic configuration of nearest noble gas.
To determine: The Lewis structure of the molecule
Answer to Problem 85E
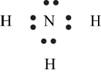
Explanation of Solution
The first step in determining the Lewis structure is to determine the number of valence electrons. The atomic number of nitrogen
The valence electron of nitrogen is 5
The atomic number of hydrogen is 1 and its electronic configuration is,
The valence electron of hydrogen is 1
The molecule
The skeletal structure of
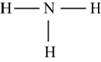
Figure 6
Each hydrogen atom requires one electron to complete the octet whereas nitrogen requires three electrons to complete the octet. In the molecule
The Lewis structure of

Figure 7
(f)
Interpretation: The Lewis structure is to be drawn for the given molecules.
Concept introduction: The Lewis structure is also known as dot structure. This structure depicts the bonding between atoms and the lone pairs of electrons if exists.
The octet rule states that atoms or molecules gain or lose electrons to get the electronic configuration of nearest noble gas.
To determine: The Lewis structure of the molecule
Answer to Problem 85E
Explanation of Solution
The first step in determining the Lewis structure is to determine the number of valence electrons. The atomic number of oxygen is 8 and its electronic configuration is,
The valence electron of oxygen is 6
The atomic number of hydrogen is 1 and its electronic configuration is,
The valence electron of hydrogen is 1
The molecule
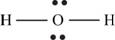
The skeletal structure of
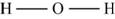
Figure 8
Each hydrogen atom requires one electron to complete the octet whereas oxygen requires two electrons to complete the octet. In the molecule
The Lewis structure of

Figure 9
(g)
Interpretation: The Lewis structure is to be drawn for the given molecules.
Concept introduction: The Lewis structure is also known as dot structure. This structure depicts the bonding between atoms and the lone pairs of electrons if exists.
The octet rule states that atoms or molecules gain or lose electrons to get the electronic configuration of nearest noble gas.
To determine: The Lewis structure of the molecule
Answer to Problem 85E
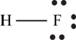
Explanation of Solution
The first step in determining the Lewis structure is to determine the number of valence electrons. The atomic number of fluorine is 9 and its electronic configuration is,
The valence electron of fluorine is 7
The atomic number of hydrogen is 1 and its electronic configuration is,
The valence electron of hydrogen is 1
The molecule
The skeletal structure of
Each hydrogen atom requires one electron to complete the octet whereas fluorine requires one electron to complete the octet. Hence, the mutual sharing of eight electrons takes place.
The Lewis structure of
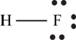
Figure 10
The Lewis dot structure is drawn to satisfy the octets of atoms. The octet rule states that elements gain or lose electrons to get the nearest noble gas configuration.
Want to see more full solutions like this?
Chapter 8 Solutions
Chemistry
- How does the bond energy of HCl(g) differ from the standard enthalpy of formation of HCl(g)?arrow_forwardConsider the following Lewis structure where E is an unknown element: What are some possible identities for element E? Predict the molecular structure (including bond angles) for this ion. (See Exercises 115 and 116.)arrow_forwardrite the Lewis structure for each of the following atoms. msp;a.He(Z=2)d.Ne(Z=10)b.Br(Z=35)e.I(Z=53)c.Sr(Z=38)f.Ra(Z=88)arrow_forward
- Lewis structures can be used to understand why some molecules react in certain ways. Write the Lewis structures for the reactants and products in the reactions described below. a. Nitrogen dioxide dimerizes to produce dinitrogen tetroxide. b. Boron trihydride accepts a pair of electrons from ammonia, forming BH3NH3. Give a possible explanation for why these two reactions occur.arrow_forwardUsing the bond energies in Table 7.2, determine the approximate enthalpy change for each of the following reactions: (a) Cl2(g)+3F2(g)2ClF3(g) (b) H2C=CH2(g)+H2(g)H3CCH3(g) (c) 2C2H6(g)+7O2(g)4CO2(g)+6H2O(g) .arrow_forwardCompare your answers from parts a and b of Exercise 69 with H values calculated for each reaction using standard enthalpies of formation in Appendix 4. Do enthalpy changes calculated from bond energies give a reasonable estimate of the actual values?arrow_forward
- Compare your answers from parts a and b of Exercise 69 of Chapter 3 with H values calculated for each reaction using standard enthalpies of formation in Appendix 4. Do enthalpy changes calculated from bond energies give a reasonable estimate of the actual values?arrow_forwardWhat is meant by a chemical bond? Why do atoms form bonds with each other? Why do some elements exist as molecules in nature instead of as free atoms?arrow_forwardWrite the Lewis symbols for each of the following ions: (a) As3 (b) I (c) Be2+ (d) O2 (e) Ga3+ (f) Li+ (g) N3arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





