
(a)
Interpretation:
Given chemical equation has to be balanced and also the oxidizing agent and reducing agent has to be identified.
Concept Introduction:
In
In redox reactions, reducing agent is the one that gets oxidized by causing reduction. These agents can be ions, elements, or even compounds. In reduction, the oxidation number decreases due to gain of electrons.
(a)
Answer to Problem K.17E
Balanced chemical equation is
Explanation of Solution
The given reaction is written as follows;
The above chemical equation has the same number of atoms of elements equal on both sides. Hence, this itself is a balanced equation.
Oxidation number of the atoms present in the above equation is indicated as follows;
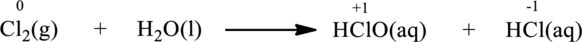
From the above equation, it is found that the oxidation state of chlorine is increased from
The oxidation state of chlorine decreases from
(b)
Interpretation:
Given chemical equation has to be balanced and also the oxidizing agent and reducing agent has to be identified.
Concept Introduction:
Refer part (a).
(b)
Answer to Problem K.17E
Balanced chemical equation is;
Oxidizing agent is
Explanation of Solution
The given reaction is written as follows;
Balancing Hydrogen atoms: In the left side of the equation there are two hydrogen atoms while on the product side only one hydrogen atom is present. Adding coefficient
Balancing Sodium atoms: In the left side of the equation there is one sodium atom while on the product side there are two sodium atoms. Adding coefficient
Balancing Chlorine atoms: In the left side of the equation there are two chlorine atoms while on the product side there is one chlorine atom. Adding coefficient
Oxidation number of the atoms present in the above equation is indicated as follows;
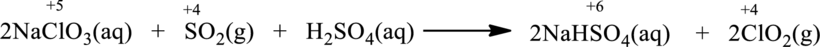
From the above equation, it is found that the oxidation state of sulfur is increased from
The oxidation state of chlorine decreases from
(c)
Interpretation:
Given chemical equation has to be balanced and also the oxidizing agent and reducing agent has to be identified.
Concept Introduction:
Refer part (a).
(c)
Answer to Problem K.17E
Balanced chemical equation is
Explanation of Solution
The given reaction is written as follows;
Balancing iodine atom: In the reactant side, there is one iodine atom while on the product side, there are two iodine atoms. Adding coefficient
Balancing copper atom: In the reactant side, there are two copper atoms while on the product side, there is one copper atom. Adding coefficient
Oxidation number of the atoms present in the above equation is indicated as follows;
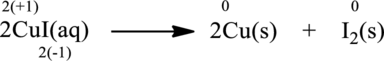
From the above equation, it is found that the oxidation state of iodine is increased from
The oxidation state of copper decreases from
Want to see more full solutions like this?
Chapter F Solutions
Chemical Principles: The Quest for Insight
- 1. Sometimes a reaction can fall in more than one category. Into what category (or categories) does the reaction of Ba(OH)2(aq) + H+PO4(aq) fit? acid-base and oxidation-reduction oxidation-reduction acid-base and precipitation precipitationarrow_forwardIf enough Li2SO4 dissolves in water to make a 0.33 M solution, explain why the molar concentration of Li+ is different from the molar concentration of Li2SO4(aq).arrow_forwardUse the appropriate tables to calculate H for (a) the reaction between MgC03(s) and a strong acid to give Mg2+(aq), CO2(g), and water. (b) the precipitation of iron(III) hydroxide from the reaction between iron(III) and hydroxide ions.arrow_forward
- Write the net ionic equation for the reaction, if any, that occurs on mixing (a) solutions of sodium hydroxide and magnesium chloride. (b) solutions of sodium nitrate and magnesium bromide. (c) magnesium metal and a solution of hydrochloric acid to produce magnesium chloride and hydrogen. Magnesium metal reacting with HCl.arrow_forwardOne half liter (500. mL) of 2.50 M HCl is mixed with 250. mL of 3.75 M HCl. Assuming the total solution volume after mixing is 750. mL, what is the concentration of hydrochloric acid in the resulting solution? What is its pH?arrow_forwardFollow the directions of Question 21 for the following bases: (a) toluidine, C7H9N (b) strontium hydroxide (c) indol, C8H6NH (d) aqueous ammoniaarrow_forward
- 4.22 Generally, an excess of O2 is needed for the reaction Sn+O2SnO2 . What is the minimum number of moles of oxygen required to oxidize 7.3 moles of tin?arrow_forwardOne of the few industrial-scale processes that produce organic compounds electrochemically is used by the Monsanto Company to produce1,4-dicyanobutane. The reduction reaction is 2CH2CHCH+2H++2eNC(CH2)4CN The NC(CH2)4CN is then chemically reduced using hydrogen gas to H2N(CH2)6NH2, which is used in the production of nylon. What current must be used to produce 150.kg NC(CH2)4CN per hour?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





