
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
8th Edition
ISBN: 9781305387102
Author: Kreith, Frank; Manglik, Raj M.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 1.80P
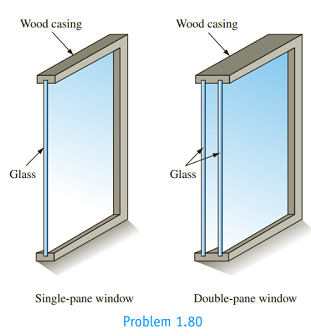
Describe and compare the modes of heat loss through the single-pane and double-pane window assemblies shown in the sketch below.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Keep in mind that both the bottom and top surfaces of the workpiece are exposed to radiation and convection. The workpiece is suspended in air in the furnace.
SOLVE STEP BY STEP IN DIGITAL FORMAT
The SOLFIES company is dedicated to welding any type of
metal, but especially aluminum parts. If the aluminum pieces
that are welded come out at 1200°C and to speed up the
cooling they are immersed in a tank full of water with a
temperature of 0°C. In this case, the heat is transferred from
the welded part to the water, causing the temperature to
begin to decrease.
Taking into account the temperature data of both the piece
and the water, in addition to the value of the constant K=
0.02, it is necessary to know how long it takes for the piece to
reach a temperature of 20°C in order to handle it and deliver
it to the customer.
MATH MODEL
T'+KT=Tm
Solve this differential equation with Euler's
numerical method.
SUPPOSE THE AMBIENT TEMPERATURE IS 20degrees CELSIUS, AND THE HOT RESERVOIR CONSISTS OF A SPHERICAL TANK WITH A RADIUS OF 4.00 m, THAT ACTS AS AN IDEAL EMITTER OF RADIATION. IF ALL THE RADIANT ENERGY EMITTED BY THE TANK COULD BE CAPTURED, WHAT IS THE AVERAGE AMOUNT OF WORK THAT COULD BE DONE EACH SECOND? ( please only answer if your 100% correct) (show work)
Chapter 1 Solutions
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
Ch. 1 - 1.1 On a cold winter day, the outer surface of a...Ch. 1 - 1.2 The weight of the insulation in a spacecraft...Ch. 1 - 1.3 A furnace wall is to be constructed of brick...Ch. 1 - 1.4 To measure thermal conductivity, two similar...Ch. 1 - To determine the thermal conductivity of a...Ch. 1 - A square silicon chip 7mm7mm in size and 0.5-mm...Ch. 1 - A cooling system is to be designed for a food...Ch. 1 - 1.80 Describe and compare the modes of heat loss...Ch. 1 - Heat is transferred at a rate of 0.1 kW through...Ch. 1 - 1.10 A heat flux meter at the outer (cold) wall of...
Ch. 1 - 1.11 Calculate the heat loss through a glass...Ch. 1 - 1.12 A wall with a thickness is made of a...Ch. 1 - 1.13 If the outer air temperature in Problem is...Ch. 1 - Using Table 1.4 as a guide, prepare a similar...Ch. 1 - 1.15 A thermocouple (0.8-mm-diameter wire) used to...Ch. 1 - Water at a temperature of 77C is to be evaporated...Ch. 1 - The heat transfer rate from hot air by convection...Ch. 1 - The heat transfer coefficient for a gas flowing...Ch. 1 - 1.19 A cryogenic fluid is stored in a...Ch. 1 - A high-speed computer is located in a...Ch. 1 - 1.21 In an experimental set up in a laboratory, a...Ch. 1 - 1.22 In order to prevent frostbite to skiers on...Ch. 1 - Using the information in Problem 1.22, estimate...Ch. 1 - Two large parallel plates with surface conditions...Ch. 1 - 1.25 A spherical vessel, 0.3 m in diameter, is...Ch. 1 - 1.26 Repeat Problem 1.25 but assume that the...Ch. 1 - Determine the rate of radiant heat emission in...Ch. 1 - 1.28 The sun has a radius of and approximates a...Ch. 1 - 1.29 A spherical interplanetary probe with a 30-cm...Ch. 1 - A spherical communications satellite, 2 m in...Ch. 1 - A long wire 0.7 mm in diameter with an emissivity...Ch. 1 - Wearing layers of clothing in cold weather is...Ch. 1 - A section of a composite wall with the dimensions...Ch. 1 - A section of a composite wall with the dimensions...Ch. 1 - Repeat Problem 1.35 but assume that instead of...Ch. 1 - 1.37 Mild steel nails were driven through a solid...Ch. 1 - Prob. 1.38PCh. 1 - 1.39 On a cold winter day, the outside wall of a...Ch. 1 - As a designer working for a major electric...Ch. 1 - 1.41 A heat exchanger wall consists of a copper...Ch. 1 - 1.43 A simple solar heater consists of a flat...Ch. 1 - A composite refrigerator wall is composed of 5 cm...Ch. 1 - An electronic device that internally generates 600...Ch. 1 - 1.47 A flat roof is modeled as a flat plate...Ch. 1 - A horizontal, 3-mm-thick flat-copper plate, 1-m...Ch. 1 - 1.49 A small oven with a surface area of is...Ch. 1 - A steam pipe 200 mm in diameter passes through a...Ch. 1 - 1.51 The inner wall of a rocket motor combustion...Ch. 1 - 1.52 A flat roof of a house absorbs a solar...Ch. 1 - Determine the power requirement of a soldering...Ch. 1 - 1.54 The soldering iron tip in Problem 1.53...Ch. 1 - Prob. 1.55PCh. 1 - A pipe carrying superheated steam in a basement at...Ch. 1 - Draw the thermal circuit for heat transfer through...Ch. 1 - 1.60 Two electric resistance heaters with a 20 cm...Ch. 1 - 1.63 Liquid oxygen (LOX) for the space shuttle is...Ch. 1 - The interior wall of a large, commercial walk-in...Ch. 1 - 1.67 In beauty salons and in homes, a ubiquitous...Ch. 1 - The heat transfer coefficient between a surface...Ch. 1 - The thermal conductivity of fibreglass insulation...Ch. 1 - 1.71 The thermal conductivity of silver at 212°F...Ch. 1 - 1.72 An ice chest (see sketch) is to constructed...Ch. 1 - Estimate the R-values for a 5-cm-thick fiberglass...Ch. 1 - A manufacturer in the United States wants to sell...Ch. 1 - Referring to Problem 1.74, how many kilograms of...Ch. 1 - 1.76 Explain a fundamental characteristic that...Ch. 1 - 1.77 Explain each in your own words. (a) What is...Ch. 1 - What are the important modes of heat transfer for...Ch. 1 - 1.79 Consider the cooling of (a) a personal...Ch. 1 - Describe and compare the modes of heat loss...Ch. 1 - A person wearing a heavy parka is standing in a...Ch. 1 - Discuss the modes of heat transfer that determine...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- An annealing furnace is at a temperature of 460 °C, where a pool of water is used to cool the heated products.is used. Each product is cylindrical, with a diameter of 25 mm and a length of 250 mm. Radiation Assuming that the coefficient is 0.85, the amount of heat transfer while the product is immersed in the water pool calculate note:this is the question, if there is missing information, take a number according to your mind and specifyarrow_forwardWrite legibly, provide given, manual step by step solution, and diagram for below problem. The filament of a 100-watt bulb maybe considered a black body radiating into a black enclosure at 80 deg C. Considering only radiation, calculate the filament temperature in deg C if the filament is 0.10mm and the length is 6cm. A. 2514 B. 2837 C. 3122 D. 2674arrow_forwardDescribe and explain Heat gain through glass. (REFRIGERATION AND AIR-CONDITIONING)arrow_forward
- A microwave dinner has the instructions listed below. In this problem, you will explain how following the instructions affect the heat transfer into the meal. (The meal starts off frozen, and is covered with a thin piece of plastic "film" or covering when it is taken out of the box. The vegetables are separate from the beef and potatoes.) Instructions1. Cut slit in film over vegetables.2. Microwave on high 4 minutes3. Turn back film from beef and potatoes. Stir beef and potatoes. Replace film. Return tray to microwave oven4. Microwave on high 3 minutes.5. Let stand 2 minutes in microwave oven., Stir beef and potatoes.a. How does each of the steps above (cut slit in film, stir beef and potatoes, etc) affect the heat transfer to the food? Make sure to consider conduction, convection, and radiation where appropriate. b. The instructions ask you to stir the beef and potatoes, but not the vegetables. From this, what can you tell about how each type of food receives and transfers heat?…arrow_forwardCalculate the total heat gains of the window below. Dimension: 9' by 15' Materials: 1" insulating glass, 1/4" lites, clear Note: SC = 0.38 due to blinds Orientation: South Time: When SHGC peaks for the South orientation on July 21, 40 N Temperature difference between design and outdoor: 33 Farrow_forwardProblem 3. If sufficient data are provided, complete the blank (non-gray) cells in the following table of properties of water (steam). In the last column describe the condition of water (steam) as compressed liquid, saturated liquid, saturated vapor, two-phase mixture, superheated vapor, or insufficient information, and, if applicable, give the quality (for two-phase mixture). A B C D E P, kPa T, C v, m'/kg h, kJ/kg Condition description and quality, if applicable 200 270.28 130 1600 70 800 120 0.2000 0.2350 2950.4arrow_forward
- A glass window pane and a pyrex window pane are held together as shown below. Heat is flowing through the combination from the hot side to the cold side. The temperatures of the hot surface and the cold surface are shown, with the temperature of the surface between them. The glass pane has a thickness of 3.00 mm. Find the thickness of the pyrex pane. (The cross-sectional areas are not needed.) Thermal conductivity of glass kglass = 0.800 J/s-m-C° Thermal conductivity of pyrex: kpyrex = 1.15 J/s-m-C° pyrex glass heat heat 3.00 mm T, = 38 °c T3 = 20° c %3D T2 = 23.4°Carrow_forwardWhat is experimental stress analysis? Methods used in experimental stress analysis Explain the Photoelasticity method with its principles by writing their names.arrow_forwardReview Conceptual Example 5 before attempting this problem. To illustrate the effect of ice on the aluminum cooling plate, consider the drawing shown here and the data contained therein. Ignore any limitations due to significant figures. (a) Calculate the heat per second per square meter that is conducted through the ice-aluminum combination. (b) Calculate the heat per second per square meter that would be conducted through the aluminum if the ice were not present. Notice how much larger the answer is in (b) as compared to (a). (a) Number i (b) Number i -10.0°C Units Units Ice -0.0050 m Aluminum -25.0°C 0.0015 marrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning

Principles of Heat Transfer (Activate Learning wi...
Mechanical Engineering
ISBN:9781305387102
Author:Kreith, Frank; Manglik, Raj M.
Publisher:Cengage Learning
Understanding Conduction and the Heat Equation; Author: The Efficient Engineer;https://www.youtube.com/watch?v=6jQsLAqrZGQ;License: Standard youtube license