
(a)
Interpretation:
The complete, detailed mechanism for the given reaction is to be drawn and the product is to be predicted.
Concept introduction:
A weak acid can add to an
Answer to Problem 11.11P
The complete mechanism of the given addition reaction is
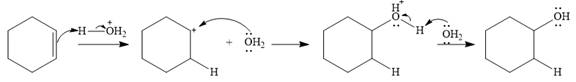
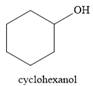
The product of the reaction is
Explanation of Solution
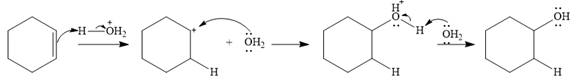
The given addition reaction is
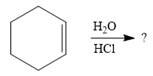
Water is a weak acid and does not add to the alkene in neutral conditions. In the presence of the strong acid HCl, water is protonated to
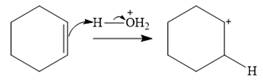
This is the electrophilic addition step. In the next step, a molecule of water acts as a nucleophile and attacks the carbocation to form protonated alcohol.
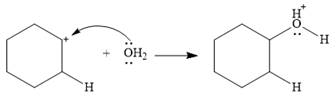
Final deprotonation by another molecule of water gives the final product, cyclohexanol.
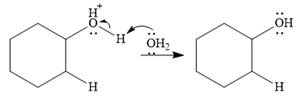
Thus, the complete mechanism for the reaction can be drawn as
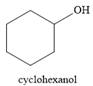
And the product of the reaction is cyclohexanol
Weak Bronsted acids can add to an alkene in the presence of a strong acid.
(b)
Interpretation:
The complete, detailed mechanism for the given reaction is to be drawn and the product is to be predicted.
Concept introduction:
A weak acid can add to an alkene in the presence of a strong acid. Because of the leveling effect, the protonated form of a weak acid is the strongest acid that can exist, so the strong acid protonates the weak acid. This protonated form of the weak acid is a good electrophile because of the positive charge. The double bond in the alkene is an electron-rich region and behaves as a nucleophile. The
Answer to Problem 11.11P
The complete mechanism for the given addition reaction is
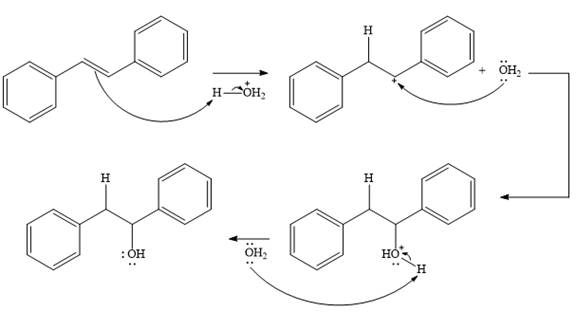
The product of the reaction is
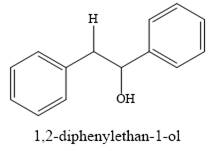
Explanation of Solution
The given addition reaction is
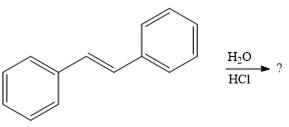
In the presence of HCl, the weak acid
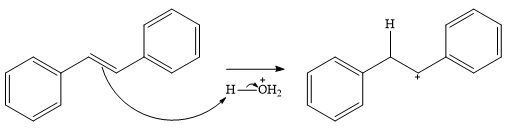
In the next step, a molecule of water acts as a nucleophile and adds to the carbocation to form a protonated alcohol.
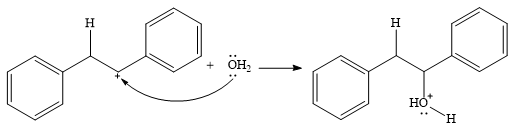
In the final step, another molecule of water deprotonates to give the final product,
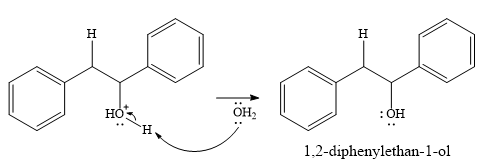
Thus, the complete mechanism for this addition reaction can be drawn as
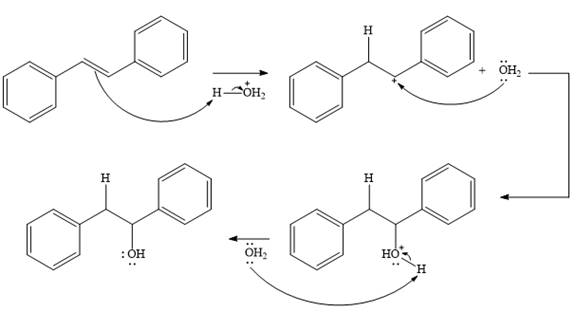
And the product of the reaction is

Weak Bronsted acids can add to an alkene in the presence of a strong acid.
(c)
Interpretation:
The complete, detailed mechanism for the given reaction is to be drawn and the product is to be predicted.
Concept introduction:
A weak acid can add to an alkene in the presence of a strong acid. Because of the leveling effect, the protonated form of a weak acid is the strongest acid that can exist, so the strong acid protonates the weak acid. This protonated form of the weak acid is a good electrophile because of the positive charge. The double bond in the alkene is an electron-rich region and behaves as a nucleophile. The
Answer to Problem 11.11P
The complete mechanism for the given addition reaction is
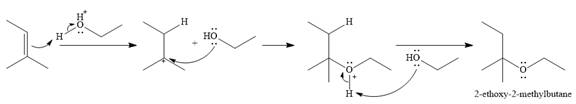
The product of the reaction is
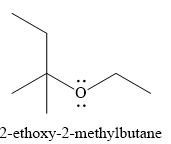
Explanation of Solution
The given reaction is
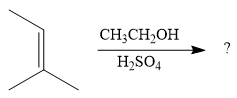
In the presence of a strong acid
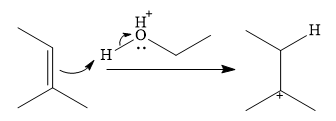
In the next step, a molecule of ethanol will act as a nucleophile and form a bond with the carbocation, using a lone pair on oxygen.
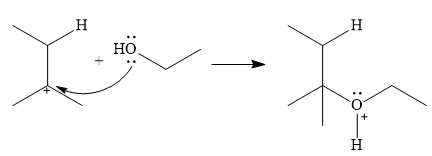
This results in the formation of protonated ether, which is deprotonated by another molecule of ethanol in the final step.
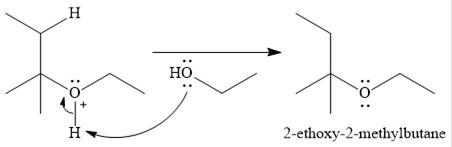
Thus, the complete mechanism for this addition reaction can be drawn as
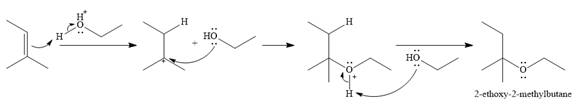
And the product of the reaction is
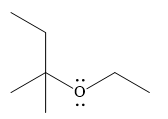
Weak Bronsted acids can add to an alkene in the presence of a strong acid.
(d)
Interpretation:
The complete, detailed mechanism for the given reaction is to be drawn and the product is to be predicted.
Concept introduction:
A weak acid can add to an alkene in the presence of a strong acid. Because of the leveling effect, the protonated form of a weak acid is the strongest acid that can exist, so the strong acid protonates the weak acid. This protonated form of the weak acid is a good electrophile because of the positive charge. The double bond in the alkene is an electron-rich region and behaves as a nucleophile. The
Answer to Problem 11.11P
The complete mechanism for the reaction is
The product of the reaction is
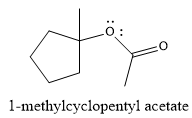
Explanation of Solution
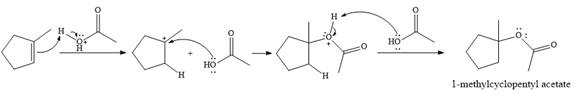
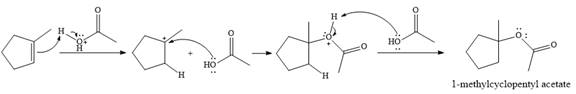
The given reaction is
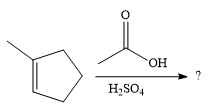
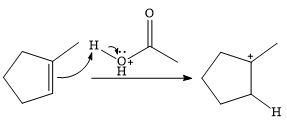
In the presence of
In the next step, a molecule of acetic acid will act as a nucleophile using a lone pair on OH oxygen to form a bond with the carbocation. The result is a protonated form of the ester product.
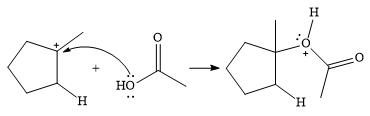
Final deprotonation by another molecule of acetic acid will give the product.
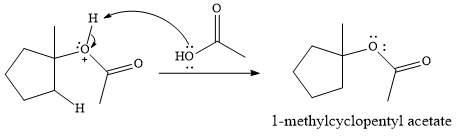
Thus, the complete mechanism for the reaction can be drawn as
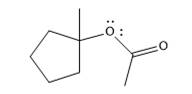
The product of the reaction is
Weak Bronsted acids can add to an alkene in the presence of a strong acid.
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Draw the complete, detailed mechanism and the products for each of the following reactions.arrow_forwardprovide the detailed mechanism for the given reactions. Do not skip any steparrow_forwardPredict the major product of each of the reactions shown here and provide the complete, detailed mechanism.arrow_forward
- The reaction shown here is an example of the Favorskii reaction, which involves an R¯ leaving group in a nucleophilic addition-elimination reaction. (a) Draw the complete, detailed mechanism for this reaction and explain why R can act as a leaving NaOH H2O group. (b) Suggest how you can synthesize an ester from cyclopropanone using only this reaction.arrow_forwardDraw a complete, detailed mechanism for the following reaction. A key intermediate is provided.arrow_forwardDraw the complete, detailed mechanism for each of the following reactions ( (b) NaOH ? U NaOH ?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





