
Concept explainers
(a)
Interpretation:
The reaction and complete, detailed mechanism for the reaction is to be drawn.
Concept introduction:
A strong Bronsted acid such as
Answer to Problem 11.27P
The overall reaction for the given compound is
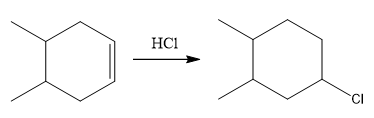
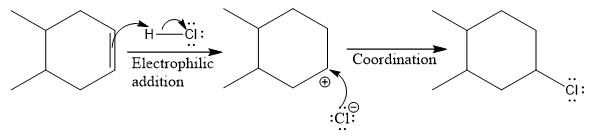
The complete mechanism for the above reaction is
Explanation of Solution
The structure for the given compound
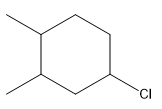
The compound shown above is the alkyl halide compound. This compound is formed by an electrophilic addition reaction of alkene with a strong Bronsted acid
The overall reaction for the given compound is

The above reaction completes in two steps. In the first step, the electrophilic addition step, the proton is added to one carbon with C=C bond, forming the carbocation intermediate. The second step is the coordination step in which the nucleophile attacks the carbocation to yield the product.
The complete mechanism for the above reaction is

The reaction and complete, detailed mechanism for the reaction is drawn on the basis of the mechanism of electrophilic addition reaction of alkenes with strong acid.
(b)
Interpretation:
The reaction and complete, detailed mechanism for the reaction is to be drawn.
Concept introduction:
Alkyl halide is the product of the electrophilic addition reaction of the alkene with a strong Bronsted acid such as
A strong Bronsted acid such as
Answer to Problem 11.27P
The overall reaction for the given compound is
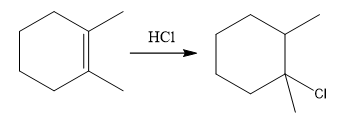
The complete mechanism for the above reaction is
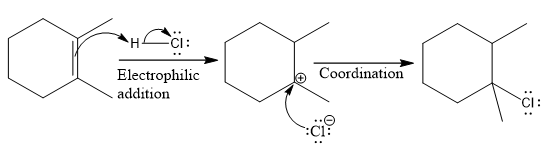
Explanation of Solution
The structure for the given compound
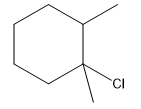
The compound shown above is the alkyl halide compound. This compound is formed by an electrophilic addition reaction of an alkene with a strong Bronsted acid
The overall reaction for the given compound is

The above reaction completes in two steps. In the first step, the electrophilic addition step, the proton is added to one carbon with C=C bond, forming the carbocation intermediate. The second step is the coordination step in which the nucleophile attacks the carbocation to yield the product.
The complete mechanism for the above reaction is

The reaction and complete, detailed mechanism for the reaction is drawn on the basis of the mechanism of electrophilic addition reaction of alkenes with strong acid.
(c)
Interpretation:
The reaction and complete, detailed mechanism for the reaction is to be drawn.
Concept introduction:
Alkyl halide is the product of the electrophilic addition reaction of the alkene with a strong Bronsted acid such as
A strong Bronsted acid such as
Answer to Problem 11.27P
The overall reaction for the given compound is
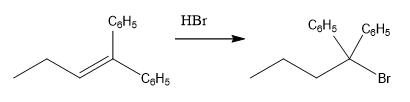
The complete mechanism for the above reaction is
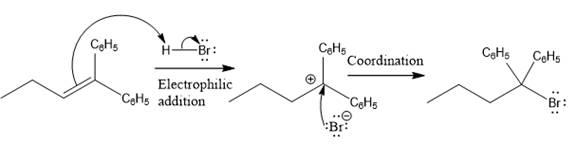
Explanation of Solution
The structure for the given compound
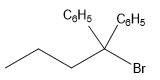
The compound shown above is the alkyl halide compound. This compound is formed by an electrophilic addition reaction of alkene with a strong Bronsted acid
The overall reaction for the given compound is

The above reaction completes in two steps. In the first step, the electrophilic addition step, the proton added to the internal carbon, forming the the more stable carbocation intermediate, which is tertiary and resonance stabilized. The second step is the coordination step in which the nucleophile attacks the carbocation to yield the product.
The complete mechanism for the above reaction is
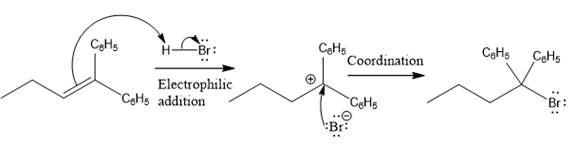
The reaction and complete, detailed mechanism for the reaction is drawn on the basis of the mechanism of electrophilic addition reaction of alkenes with strong acid.
(d)
Interpretation:
The reaction and complete, detailed mechanism for the reaction is to be drawn.
Concept introduction:
Alkyl halide is the product of the electrophilic addition reaction of the alkene with a strong Bronsted acid such as
A strong Bronsted acid such as
Answer to Problem 11.27P
The overall reaction for the given compound is
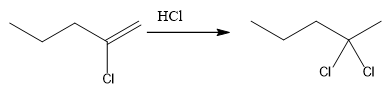
The complete mechanism for the above reaction is
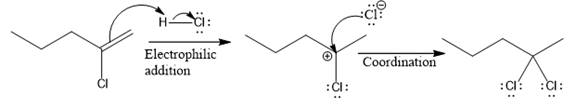
Explanation of Solution
The structure for the given compound
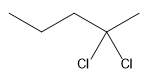
The compound shown above is the alkyl halide compound. This compound is formed by an electrophilic addition reaction of alkene with a strong Bronsted acid
The overall reaction for the given compound is

The above reaction completes in two steps. In the first step, the electrophilic addition step, the proton is added to the internal carbon, forming the the more stable carbocation intermediate, which is tertiary. The second step is the coordination step in which the nucleophile attacks the carbocation to yield the product.
The complete mechanism for the above reaction is
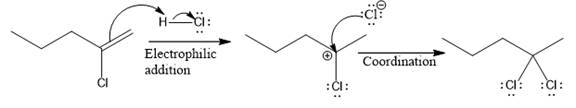
The reaction and complete, detailed mechanism for the reaction is drawn on the basis of the mechanism of electrophilic addition reaction of alkenes with strong acid.
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Provide a complete, detailed mechanism for the reaction CH;CH,OH shown here.arrow_forwardProvide the complete mechanism for the reaction. Please include appropriate arrows, intermediates, and formal charges.arrow_forwardProblem (a) Why is the following reaction a poor method for the synthesis of t propyl ether? (b) What would be the major product from this reaction? (c) Propose a better synthesis of t-butyl propyl ether. does not CH, give ÇH, CH,CH,CH,-0:- *Na + CH,-Ċ-Br CH;-Ç-0-CH,CH, CH, CH, sodium propoxide 1-butyl bromide r-butyl propyl ctherarrow_forward
- In the acid-catalyzed aromatic alkylation involving 1-methylcyclohexene and benzene, two isomeric products are possible, but only one is formed, as shown here. Draw the complete mechanism that leads to each product, and explain why only one isomer is formed.arrow_forwardDraw the complete, detailed mechanism for each of the following reactions and predict the major product(s). (a) (b) Br,/HO ? Br,/H H2O H2Oarrow_forward2) Provide a detailed mechanism for the addition reaction shown below. State the regiochemistry and explain why it's unusual. SCH3 HBr Br SCH3 EN Please be sure to include all structures (use line angle notation or perspective diagrams as appropriate to illustrate the stereochemistry of the process), resonance forms, intermediates, transition states, curved arrows, formal charges, or lone pairs as necessary. Please redraw the substratearrow_forward
- Can you please help with the following organic chemistry reaction (see attached image) Provide the mechanism involved in the reaction and what the major product(s) would be. Thank youarrow_forward3) Show a complete mechanism and predict the product for the reaction shown below. Br₂ / H₂O Please provide perspective diagrams (wedge/dash notation), resonance forms, curved arrows, formal charges, and lone pairs as need for all species involved. With reference to the intermediate(s) involved, comment on both the regiochemistry and the stereochemistry of the process.arrow_forwardORGANIC CHEMISTRY: Is this mechanism for dibenzalacetone formation complete? arrows and electrons, and formal charges?. If no, please provide the complete mechanism. From this mechanism in the picture, identify the intermediate benzalacetone.arrow_forward
- Can someone explain to me the mechanism for the reaction? I don't know what type of reaction this is and how they add together to create the product. I do understand the actual product is correct because having all substituents equitorial is favorable.arrow_forwardOchem... What is the major product for the following three-step reaction sequence? Also, provide the structures for the major organic products formed in each step. See attached imagearrow_forward2) Provide a detailed mechanism for the addition reaction shown below. State the regiochemistry and explain why it's unusual. Please be sure to include all structures (use line angle notation or perspective diagrams as appropriate to illustrate the stereochemistry of the process), resonance forms, intermkediates, transition states, curved arrows, formal charges, or lone pairs as necessary. Please don't cheat. Br HBr SCH3 SCH + ENarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





