
Concept explainers
(a)
Interpretation:
The class of given compound is to be identified.
Concept introduction:
The
Answer to Problem 44E
The class of compound
Explanation of Solution
The given organic compound is
Alcohol is the class of organic compounds in which an alkyl group is attached to hydroxyl group. The general formula of alcohols is
Therefore, the class of compound
The class of given has been rightfully identified.
(b)
Interpretation:
The class of given compound is to be identified.
Concept introduction:
The functional groups are atoms or the group of atoms that acts as substituents in the molecules and are responsible for their characteristic reactions. The functional group containing oxygen atoms are alcohol group, carboxylic acid group, aldehyde group, ketone group, phenols, ethers, and their derivatives.
Answer to Problem 44E
The class of compound
Explanation of Solution
The given organic compound is
Ether is the derivatives of alcohol. The general formula of alcohols is shown below.
When the hydrogen of the alcohol is replaced by another alkyl group ether is formed. The general formula of ether is shown below.
Therefore, the class of compound
The class of given compound has been rightfully identified.
(c)
Interpretation:
The class of given compound is to be identified.
Concept introduction:
The functional groups are atoms or the group of atoms that acts as substituents in the molecules and are responsible for their characteristic reactions. The functional group containing oxygen atoms are alcohol group, carboxylic acid group, aldehyde group, ketone group, phenols, ethers, and their derivatives.
Answer to Problem 44E
The class of the given compound is amide.
Explanation of Solution
The structure of the given compounds is shown below.
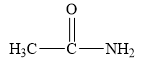
Figure 1
Amides are the derivatives of carboxylic acid. The general structure of carboxylic acid is shown below.
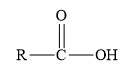
Figure 2
When the hydroxyl group of carboxylic acid is replaced by
Therefore, the class of the given compound is amide.
The class of given has been rightfully identified.
(d)
Interpretation:
The class of given compound is to be identified.
Concept introduction:
The functional groups are atoms or the group of atoms that acts as substituents in the molecules and are responsible for their characteristic reactions. The functional group containing oxygen atoms are alcohol group, carboxylic acid group, aldehyde group, ketone group, phenols, ethers, and their derivatives.
Answer to Problem 44E
The class of the given compound is ketone.
Explanation of Solution
The structure of the given organic compound is shown below.
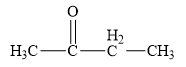
Figure 3
The carbonyl compounds are the compounds that contains
Therefore, the class of the given compound is ketone.
The class of given has been rightfully identified.
(e)
Interpretation:
The class of given compound is to be identified.
Concept introduction:
The functional groups are atoms or the group of atoms that acts as substituents in the molecules and are responsible for their characteristic reactions. The functional group containing oxygen atoms are alcohol group, carboxylic acid group, aldehyde group, ketone group, phenols, ethers, and their derivatives.
Answer to Problem 44E
The class of the given compound is ester.
Explanation of Solution
The structure of the given compounds is shown below.
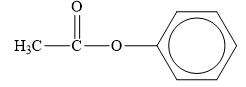
Figure 4
Esters are the derivatives of carboxylic acid. The general structure of carboxylic acid is shown below.
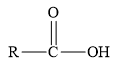
Figure 2
When the hydrogen of the hydroxyl group of carboxylic acid is replaced by an alkyl or aryl group, then ester is formed.
Therefore, the class of the given compound is ester.
The class of given has been rightfully identified.
(f)
Interpretation:
The class of given compound is to be identified.
Concept introduction:
The functional groups are atoms or the group of atoms that acts as substituents in the molecules and are responsible for their characteristic reactions. The functional group containing oxygen atoms are alcohol group, carboxylic acid group, aldehyde group, ketone group, phenols, ethers, and their derivatives.
Answer to Problem 44E
The class of given compound is phenol.
Explanation of Solution
The structure of the given organic compound is shown below.
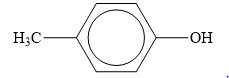
Figure 5
When an
Therefore, the class of given compound is phenol.
The class of given has been rightfully identified.
(g)
Interpretation:
The class of given compound is to be identified.
Concept introduction:
The functional groups are atoms or the group of atoms that acts as substituents in the molecules and are responsible for their characteristic reactions. The functional group containing oxygen atoms are alcohol group, carboxylic acid group, aldehyde group, ketone group, phenols, ethers, and their derivatives.
Answer to Problem 44E
The class of the given compound is organic halide.
Explanation of Solution
The structure of the given organic compound is shown below.
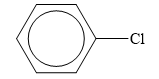
Figure 6
Halogen is the elements that belong to the seventeenth group of the periodic table. When a halogen is combined to a hydrocarbon, then organic halides are formed. Chlorine is halogen.
Therefore, the class of the given compound is organic halide.
The class of given has been rightfully identified.
(h)
Interpretation:
The class of given compound is to be identified.
Concept introduction:
The functional groups are atoms or the group of atoms that acts as substituents in the molecules and are responsible for their characteristic reactions. The functional group containing oxygen atoms are alcohol group, carboxylic acid group, aldehyde group, ketone group, phenols, ethers, and their derivatives.
Answer to Problem 44E
The class of the given compound is alcohol.
Explanation of Solution
The structure of the given compounds is shown below.
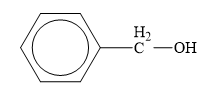
Figure 7
Alcohol is the class of organic compounds in which an alkyl group is attached to hydroxyl group. The general formula of alcohols is
Therefore, the class of the given compound is alcohol.
The class of given has been rightfully identified.
Want to see more full solutions like this?
Chapter 19 Solutions
Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- Following is the structural formula of acetylsalicylic acid, better known by its common name aspirin. (a) Name the two oxygen-containing functional groups in aspirin. (b) What is the molecular formula of aspirin?arrow_forwardWrite two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.(a) 2-butene reacts with chlorine.(b) benzene burns in air.arrow_forward(a) The compound given below had the following IUPAC name and structural formula dibromocyclopentane C3H6CHBrCHBr (i) What type of isomerism is possible in the organic compound? (ii) Draw all the pairs of possible isomers and name them.arrow_forward
- Drawthe characteristic functional group of FOUR of the following six families of organic compounds: Alcohol, Amine, Aldehyde, Ketone, Carboxylic acid, or Ester.arrow_forwardDraw the structural formulas for the following compounds. Include all the bonds to hydrogen atoms. Be sure to answer both parts. (a) 1,4-dichloro-2-ethylbenzene: (b) 2-ethyl-1,3-dimethylbenzene:arrow_forwardGive a definition and an example for each class of organic compounds.(a) alkane (b) alkene (c) alkyne(d) alcohol (e) ether (f) ketonearrow_forward
- Write the chemical formula and Lewis structure of the following, each of which contains five carbon atoms:(a) an alkane(b) an alkene(c) an alkynearrow_forwardWhat is the complete IUPAC name for each of the following molecules? (a) Br (b) CI (c) F (d) Ph OCH3 CIarrow_forwardPentane and pentene. (a) Are isomers because they have the same molecular formula. (b) Are isomers because they have the same number of carbon atoms. (c) Are not isomers because they have different molecular formulas (d) Are not isomers because they have different namesarrow_forward
- (a) Which of the following compounds, if any, is an ether? (b) Which compound, If any, is an alcohol? (c) Which com- pound, if any, would produce a basic solution if dissolved in water? (Assume solubility is not a problem). (d) Which compound, if any, is a ketone? (e) Which compound, if any, is an aldehyde? () Н,С—CH;—он Н (ii) H;C-Ñ-CH,CH=CH2 (ii) o (iv) (v) CH;CH,CH,CH2CHO (vi) CH3C=CCH,COOHarrow_forwardClassify each of he following hydrocarbons as alkanes, alkenes, or alkynes. (a) C6H14 (b) C3H4 (c) C9H18arrow_forwardTRUE OR FALSE (a) A functional group is a group of atoms in an organic molecule that undergoes a predictable set of chemical reactions. (b) The functional group of an alcohol, an aldehyde, and a ketone have in common the fact that each contains a single oxygen atom. (c) A primary alcohol has one —OH group, a secondary alcohol has two —OH groups, and a tertiary alcohol has three —OH groups. (d) There are two alcohols with the molecular formula C3H8O. (e) There are three amines with the molecular formula C3H9N. (f) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (g) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (h) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic…arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





