
Concept explainers
(a)
Interpretation:
The structure of the ether with the name “diisopropyl ether” is to be stated.
Concept introduction:
The systematic naming of organic compound is given by
Answer to Problem 54E
The structure of the ether with the name “diisopropyl ether” is
Explanation of Solution
The given name “diisopropyl ether” is the common name of ether. The diisopropyl in the name represents the presence of two isopropyl groups on both sides of oxygen in the ether structure.
The structure of “diisopropyl ether” is shown below.
The structure of the “diisopropyl ether” is
(b)
Interpretation:
The structure of the ether with the name “diphenyl ether” is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is given by some set of rules in order to give each compound its unique name. Common names are also given to the organic compounds. Common names of organic compounds ethers is given by writing the parent chain name as prefix and function group as the suffix. The prefix for hydrocarbon parent chain is “yl” in the end of name for parent chain. The suffix for the functional group is “ether”.
Answer to Problem 54E
The structure of the ether with the name “diphenyl ether” is shown below.
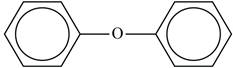
Explanation of Solution
The given name “diphenyl ether” is the common name of ether. The diphenyl in the name represents the presence of two phenyl groups on both sides of oxygen in the ether structure.
The structure of “diphenyl ether” is shown below.
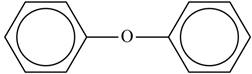
Figure 1
The structure of the “diphenyl ether” is shown in Figure 1.
(c)
Interpretation:
The structure of the ether with the name “ethyl propyl ether” is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is given by some set of rules in order to give each compound its unique name. Common names are also given to the organic compounds. Common names of organic compounds ethers is given by writing the parent chain name as prefix and function group as the suffix. The prefix for hydrocarbon parent chain is “yl” in the end of name for parent chain. The suffix for the functional group is “ether”.
Answer to Problem 54E
The structure of the ether with the name “ethyl propyl ether” is
Explanation of Solution
The given name “ethyl propyl ether” is the common name of ether. The ethyl and propyl in the name represents the presence one ethyl and one propyl group around oxygen in the ether structure.
The structure of “ethyl propyl ether” is shown below.
The structure of the “ethyl propyl ether” is
(d)
Interpretation:
The structure of the ether with the name “methyl phenyl ether” is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is given by some set of rules in order to give each compound its unique name. Common names are also given to the organic compounds. Common names of organic compounds ethers is given by writing the parent chain name as prefix and function group as the suffix. The prefix for hydrocarbon parent chain is “yl” in the end of name for parent chain. The suffix for the functional group is “ether”.
Answer to Problem 54E
The structure of the ether with the name “methyl phenyl ether” is shown below.
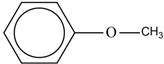
Explanation of Solution
The given name “methyl phenyl ether” is the common name of ether. The methyl and phenyl in the name represents the presence one methyl and one phenyl group around oxygen in the ether structure.
The structure of “methyl phenyl ether” is shown below.
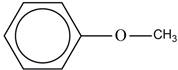
Figure 2
The structure of the “methyl phenyl ether” is shown in Figure 2.
Want to see more full solutions like this?
Chapter 19 Solutions
Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- Complete following conversions: (a) Acetic acid to methylamine (b) Acetylene to aldehydearrow_forwardGive an example reaction that would yield the following products. Name the organic reactant and product in each reaction. (a)alkane (b)monohalogenated alkanearrow_forwardProvide the IUPAC name for each of the following ketones. (a) (b) (c) (d) NO2 CI O,N' `NO2arrow_forward
- State the different kinds of arrows used in describing organic reactions ?arrow_forwardWrite two complete balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.(a) propanol is converted to dipropyl ether(b) propene is treated with water in dilute acid.arrow_forwardProvide the IUPAC name for each of the following compounds. (a) (b) (c) NH, 0 NH2 OH NH2 OHarrow_forward
- What is the IUPAC name of this organic molecule?arrow_forwardName the following molecules. (a) (b) (c) (d)arrow_forwardIn each of the following classes of organic compounds, write a molecule with a total of 16 carbons (by express formula) with at least one aromatic group and one alkyl group in its structure. Name each structure you write according to the IUPAC system. a) Write down and name a dithiol molecule. b) Write down a lactone molecule and name it.arrow_forward
- Write a balanced equation for the complete combustion for each of the following. (a) methane (b) propane (c) hexanearrow_forwardDraw the condensed structure of the compounds formedby condensation reactions between (a) benzoic acid andethanol, (b) ethanoic acid and methylamine, (c) acetic acidand phenol. Name the compound in each case.arrow_forwardThe incorrect information is given in the choice of a) Aromatic compounds have a special type of unsaturation and are the derivatives of fused benzene rings. b) Compounds with no multiple bonds (covalent) between carbon atoms are called unsaturated compounds. c) Alkanes having 1-4 C atoms are all gaseous compounds. d) Alkyl groups are obtained by removing a hydrogen atom from an alkane. e) Ethers have the general formula of R-O-R or R-O-R' where R groups may be the same or different or aromatic.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


