
Concept explainers
(a)
Interpretation:
The polarity of the given molecule is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
The given molecule A is nonpolar.
Explanation of Solution
![]()
The given molecule is in trans form. The directions of the vectors of both the C-F bonds are equal but opposite to each other. Hence the dipole moments of both the C-F bonds get cancelled out with each other. Therefore, there is no net dipole moment.
Dipole moment on this molecule is symmetrically distributed; hence the given molecule A is nonpolar.
(b)
Interpretation:
The polarity of the given molecule is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
The given molecule B is polar.
Explanation of Solution
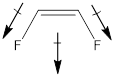
The given molecule is in cis form. The direction of vectors of both the C-F bonds is in the same direction, giving a net permanent dipole moment to the molecule.
Dipole moment on this molecule is not symmetrically distributed; hence the given molecule B is polar.
(c)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule C is nonpolar.
Explanation of Solution
![]()
In this molecule, both the C-Cl bonds are opposite to each other, so the dipole moments are cancelled out with each other. Therefore, there is no net dipole moment in this molecule.
Dipole moment on this molecule is symmetrically distributed; hence the given molecule C is nonpolar.
(d)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a.
Answer to Problem 2.41P
The given molecule D is polar.
Explanation of Solution
![]()
In this molecule, chlorine is more electronegative than the carbon atom; hence the direction of the vector of dipole moment is more towards C-Cl bond, giving a net dipole moment to the molecule.
Dipole moment on this molecule is not symmetrically distributed; hence the given molecule D is polar.
(e)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule E is polar.
Explanation of Solution
![]()
In this molecule, Chlorine is more electronegative than bromine; hence the direction of the vector of dipole moment is more towards C-Cl bond, giving a net dipole moment to the molecule.
Dipole moment on this molecule is not symmetrically distributed; hence the given molecule E is polar.
(f)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule F is nonpolar.
Explanation of Solution
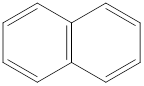
In this molecule, there is no electronegative atom present since no charge separation is taking place. So there is no net dipole moment.
Dipole moment on this molecule is symmetrically distributed; hence the given molecule F is nonpolar.
(g)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule G is polar.
Explanation of Solution
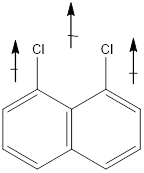
In this molecule, Chlorine is an electronegative atom, and both the C-Cl bonds are in the same direction. Therefore, the direction of the vector of dipole is moment is upward, giving a net dipole moment to the molecule.
Dipole moment on this molecule is not symmetrically distributed; hence the given molecule G is polar.
(h)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule H is nonpolar.
Explanation of Solution
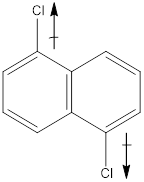
In the molecule, chlorine is an electronegative atom, and both the C-Cl bonds are in opposite direction. Therefore, the directions of the vectors of dipole moment of two C-Cl bonds get cancelled out with each other. Hence there is no net dipole moment.
Dipole moment on this molecule is symmetrically distributed; hence the given molecule H is nonpolar.
(i)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule I is polar.
Explanation of Solution
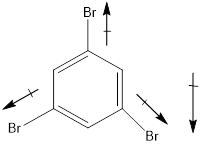
In this molecule, bromine is an electronegative atom, but one C-Br bond is in upward direction, and two C-Br bonds are in downward direction. Therefore, the net dipole moment acts in downward direction.
Dipole moment on this molecule is not symmetrically distributed; hence the given molecule I is polar.
(j)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule J is polar.
Explanation of Solution
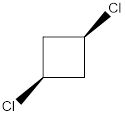
In this molecule, both the C-Cl bonds are present above the plane (that is wedge notation). Therefore, the directions of the vectors of dipole moment of both the C-Cl bonds are in the same direction, giving net dipole moment to the molecule.
Dipole moment on this molecule is not symmetrically distributed; hence the given molecule J is polar.
(k)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule K is polar.
Explanation of Solution
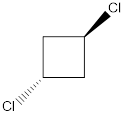
In this molecule, one C-Cl bond is present above the plane (that is, the wedge notation), and another C-Cl bond is present below the plane (that is, the dotted notation). Therefore, the directions of the vectors of dipole moment of both the C-Cl bonds are in opposite direction, which get cancelled out with each other, giving no net dipole moment to the molecule.
Dipole moment on this molecule is symmetrically distributed; hence the given molecule K is nonpolar.
(l)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule L is polar.
Explanation of Solution

In this molecule, though both the C-Cl bonds are in opposite direction, both the chlorines are present on carbon
Dipole moment on this molecule is not symmetrically distributed; hence the given molecule L is polar.
(m)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule M is nonpolar.
Explanation of Solution
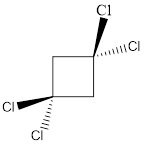
In this molecule, two C-Cl bonds are above the plane, and two C-Cl bonds are below the plane; hence the molecule has symmetry. The directions of the vectors of dipole moment of all the four C-Cl bonds are cancelled with each other, giving no net dipole moment to the molecule.
Dipole moment on this molecule is symmetrically distributed; hence the given molecule M is nonpolar.
(n)
Interpretation:
The polarity of the given molecules is to be determined.
Concept introduction:
The dipole moment of a molecule is a measure of the magnitude of its dipole. A dipole moment is a vector, which has both magnitude and direction. Bond polarity originates from bonds between atoms of different electronegativity. Symmetry of molecules also predicts the polarity of a molecule.
Answer to Problem 2.41P
Molecule N is polar.
Explanation of Solution
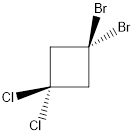
In this molecule, two C-Cl and two C-Br bonds are present. Since chlorine is more electronegative than bromine, the direction of the vector of dipole moment is towards C-Cl bonds. Therefore, there is a net dipole moment present in this molecule.
Dipole moment on this molecule is not symmetrically distributed; hence the given molecule N is polar.
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Explain why the two molecules below cannot inter-convert without breaking the portion of thedouble bond.arrow_forwardSelect ALL of the following that can be used to explain the polarities of NO2 and SeCl42- (these molecules are mentioned in earlier questions). NO2 has a permanent dipole moment or a net dipole moment greater than zero whereas SeCl42- has no net or zero dipole moment. Both molecules are symmetrical and hence nonpolar Due to their respective shapes, in NO2 the bond dipole moments reinforce each other whereas in SeCl42- they cancel out each other. NO2 has no net dipole moment and SeCl42- has a net dipole moment greater than zero. NO2 is an unsymmetrical molecule whereas SeCl42- is a symmetrical ion.arrow_forwardThe instructions are to draw the resonance hybrid structure of the molecule on the left. My answer is circled , which is wrong. Can someone explain why?arrow_forward
- A student spills a dye made predominantly of hydrogen and carbon that has several benzene rings in its structure. The dye is spilled onto the student's T-shirt. The student runs to the water tap and washes the spot with water. What result would you expect to see, and why? The dye washed off, because water is a polar substance that can bind to the polar dye. The dye washed off, because the polar water binds to the non-polar dye more strongly than the dye binds to the T-shirt. The dye did not wash off, because the polar-dye has bound strongly to the T- shirt and cannot be washed off with water. The dye did not wash off, because water is a polar substance that cannot wash the non-polar dye.arrow_forwardSelect the single best answer. For the following molecule, indicate the positive and negative ends of the dipole, using the symbol →→→→. Refer to a table of the Pauling electronegativity scale as needed. HF The arrow points to the left. The arrow points to the right.arrow_forwardis TETRACYANOETHYLENE polar or nonpolar?arrow_forward
- Examine the ungraded ball-and-stick model below to determine the three-dimensional structure of the molecule. On the corresponding 2D structure, draw one wedge bond and one dash bond over two existing bonds to indicate the same arrangement of atoms in space. The narrow part of each wedge-and-dash bond should be towards the same central carbon atom.arrow_forwardConsider this Lewis structure in the curved arrows joke. Draw the resident structure obtained by moving electrons as indicated by the curved arrows.arrow_forwardConsider the following molecule. The lone pair on the nitrogen is located in a From the list below select the correct answer that fills in the blank. O sp N it is outside of an orbital Osp2 Osp3 orbital.arrow_forward
- Select the answer that describes this molecules polarity. Consider its shape in 3 dimensionsarrow_forwardWhich ones are polar and which ones are non polar?arrow_forwardDraw each molecule with its correct geometry in 3D and indicate the direction of the molecule's overall dipole, if none label it as zero.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



