
Concept explainers
(a)
Interpretation:
The VSEPR formula and shape for
Concept Introduction:
Valence Shell Electron Pair Repulsion model predicts shape by inclusion of bond angles and most distant arrangement of atoms that leads to minimum repulsion. For the molecules that have no lone pairs around the central atom the bonded-atom unshared -pair arrangement is decided by the table as follows:
In order to determine the shape the steps to be followed are indicated as follows:
- 1. Lewis structure of molecule should be written.
- 2. The type electron arrangement around the central atom should be identified around the central atom. This essentially refers to determination of bond pairs and unshared or lone pairs around central atoms.
- 3. Then bonded-atom unshared -pair arrangement that can maximize the distance of electron pairs about central atom determines the shape.
For molecules that have lone pairs around central atom, lone pairs influence shape, because there are no atoms at the positions occupied by these lone pairs. The key rule that governs the molecular shape, in this case, is the extent of lone –lone pair repulsions are far greater than lone bond pair or bond pair-bond pair repulsions. The table that summarized the molecular shapes possible for various combinations of bonded and lone pairs are given as follows:
(a)
Answer to Problem 2E.12E
The shape for
Explanation of Solution
Total valence electrons are sum of the valence electrons on each atom along with uni-negative charge in
The skeleton structure in
These 13 electron pairs are assigned as lone pairs of each of the
Hence, the Lewis structure
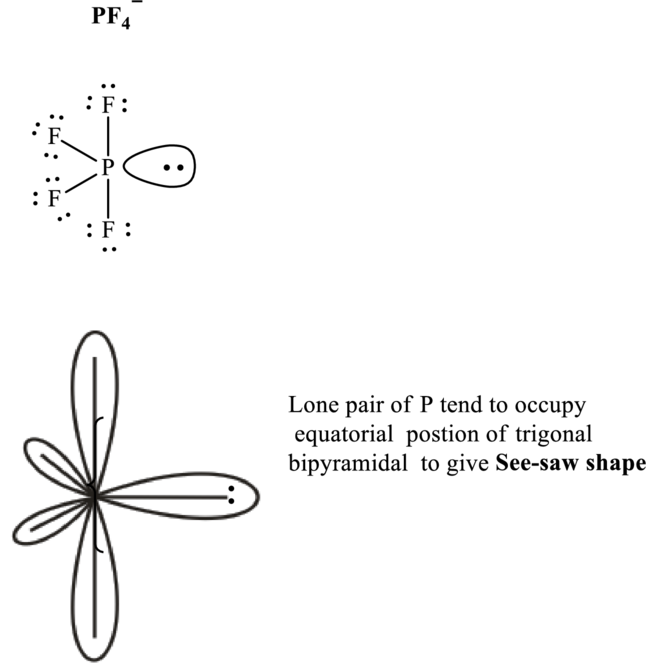
If lone pairs are represented by E, central atom with A and other attached bon pairs by X, then for any see-saw species the VSEPR formula is predicted as
It is evident that in
(b)
Interpretation:
The VSEPR formula and shape for
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 2E.12E
The shape for
Explanation of Solution
Total valence electrons are sum of the valence electrons on each chlorine and central iodine in
The skeleton structure in
These 13 electron pairs are allotted as lone pairs of each of the chlorine atoms and central iodine to satisfy respective octets. Hence, the Lewis structure and corresponding VSPER geometry in
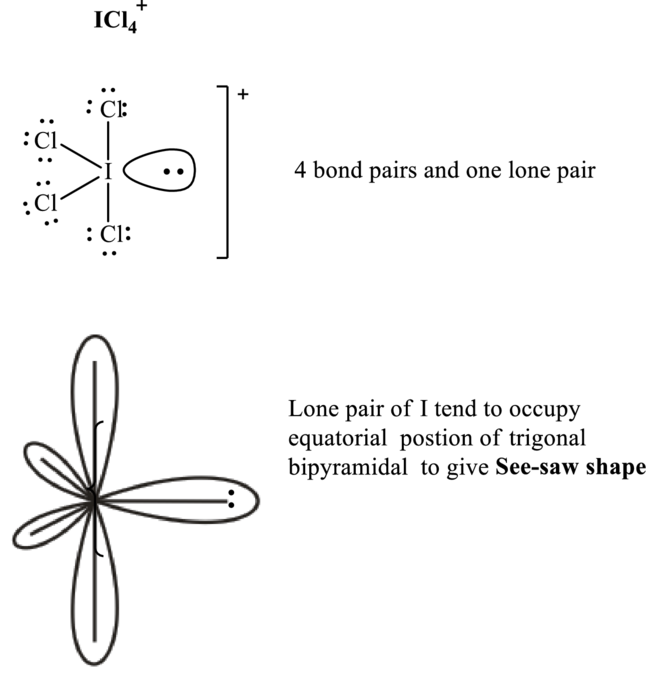
It is evident that in
Lone pairs tend to occupy the equatorial locations of trigonal plane in trigonal bi-pyramidal arrangement so as to have minimum repulsions in accordance with VSPER model. This results in see-saw shaped
(c)
Interpretation:
The VSEPR formula and shape for
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 2E.12E
The shape for
Explanation of Solution
Total valence electrons are sum of the valence electrons on each atom along with uni-negative charge in
The skeleton structure in
These 15 electron pairs are assigned as lone pairs of each of the
Hence, Lewis structure of
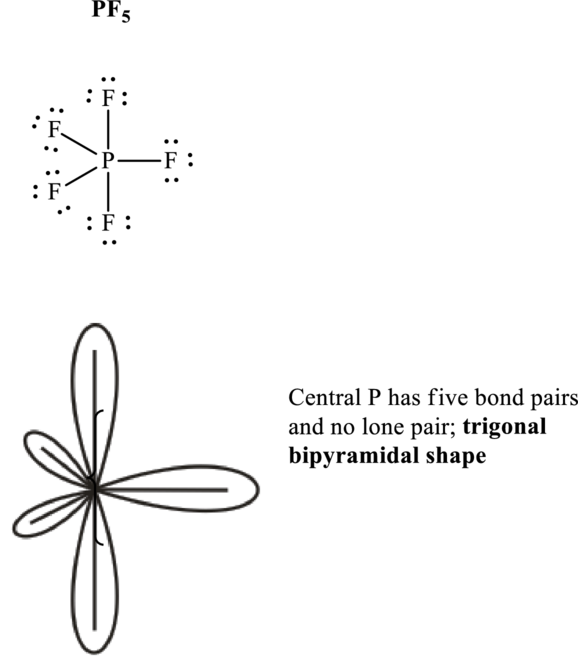
If lone pairs are represented by E, central atom with A and other attached bond pairs by X, then for any trigonal pyramidal geometry the VSEPR formula is predicted as
It is evident that in
(d)
Interpretation:
The VSEPR formula and shape for xenon terafluoride molecule has to be predicted.
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 2E.12E
The shape for xenon terafluoride molecule is square planar and corresponding VSEPR formula is
Explanation of Solution
Xenon terafluoride has
Total valence electrons are sum of the valence electrons on each oxygen atom and central
The skeleton structure in
These 14 electron pairs are allotted as lone pairs of each of the fluorine atoms and central xenon to satisfy respective octets. Thus, the Lewis structure and corresponding VSEPR geometry
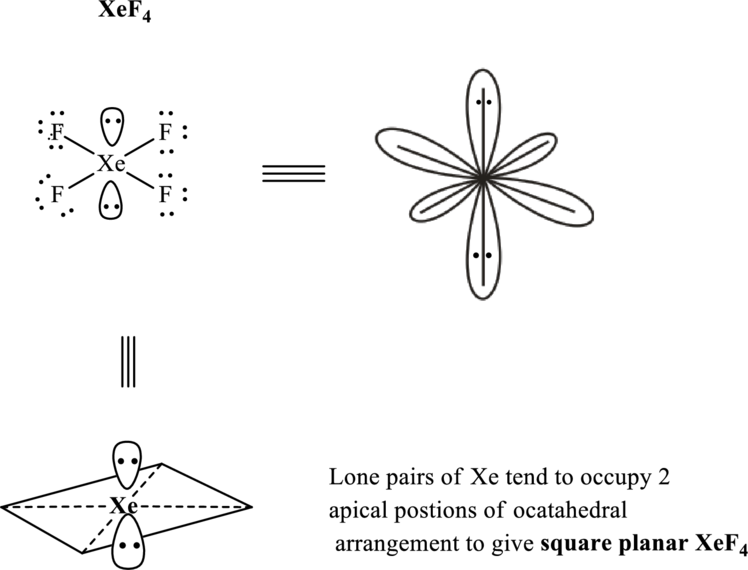
It is evident that in
If lone pairs are represented by E, central atom with A and other attached bond pairs by X, then for square planar any species the VSEPR formula is predicted as
Want to see more full solutions like this?
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Draw the Lewis structure with lowest formal charges, and determine the charge of each atom in (a) OCS; (b) NO. (C)CN−; (d) ClO−.arrow_forwardWhich of the following bonds are polar: (a) P—O; (b) S—F; (c) Br—Br; (d) O—Cl? Which is the more electronegative atom in each polar bond?arrow_forwardFrom their Lewis structures, determine the number of sand p bonds in each of the following molecules or ions:(a) CO2; (b) cyanogen, 1CN22; (c) formaldehyde, H2CO;(d) formic acid, HCOOH, which has one H and two O atomsattached to C.arrow_forward
- Like several other bonds, carbon-oxygen bonds havelengths and strengths that depend on the bond order. Draw Lewis structures for the following species, and arrange them in order of increasing carbon-oxygen bond length and then by increasing carbon-oxygen bond strength: (a) CO; (b) CO₃²⁻; (c) H₂CO;(d) CH₄O; (e) HCO₃(H attached to O).arrow_forwardDraw the Lewis structure with lowest formal charges, anddetermine the charge of each atom in (a) BF₄⁻; (b) ClNO.arrow_forwardCompounds such as NaBH4, Al(BH4)3, and LiAlH4 are complexhydrides used as reducing agents in many syntheses.(a) Give the oxidation state of each element in these compounds.(b) Write a Lewis structure for the polyatomic anion in NaBH4, and predict its shape.arrow_forward
- . Assume that the third-period element phosphorus forms a diatomic molecule, P2, in an analogous way as nitrogen does to form N2. (a) Write the electronic configuration for P2. Use [Ne2] to represent the electron configuration for the first two periods. (b) Calculate its bond order. (c) What are its magnetic properties (diamagnetic or paramagnetic)?arrow_forwardThe sulfate ion can be represented with four S-O bonds or with two S-O and two So=O bonds.(a) Which representation is better from the standpoint of formal charges?(b) What is the shape of the sulfate ion, and what hybrid orbitals of S are postulated for the σ bonding?(c) In view of the answer to part (b), what orbitals of S must be used for the π bonds? What orbitals of O?(d) Draw a diagram to show how one atomic orbital from S and one from O overlap to form a π bond.arrow_forwardBy referring only to the periodic table, select (a) the most electronegativeelement in group 6A; (b) the least electronegativeelement in the group Al, Si, P; (c) the most electronegative elementin the group Ga, P, Cl, Na; (d) the element in the group K,C, Zn, F that is most likely to form an ionic compound with Ba.arrow_forward
- Acetylene 1C2H22 and nitrogen 1N22 both contain a triplebond, but they differ greatly in their chemical properties.(a) Write the Lewis structures for the two substances. (b) Byreferring to Appendix C, look up the enthalpies of formationof acetylene and nitrogen. Which compound is more stable?(c) Write balanced chemical equations for the completeoxidation of N2 to form N2O51g2 and of acetylene to formCO21g2 and H2O1g2. (d) Calculate the enthalpy of oxidationper mole for N2 and for C2H2 (the enthalpy of formationof N2O51g2 is 11.30 kJ>mol). (e) Both N2 and C2H2 possesstriple bonds with quite high bond enthalpies (Table 8.3).Calculate the enthalpy of hydrogenation per mole for bothcompounds: acetylene plus H2 to make methane, CH4;nitrogen plus H2 to make ammonia, NH3.arrow_forward4. (a) Draw the shape of the atomic valence orbitals formed by the overlaping of two fluoride 2p atomic orbitals. (b) Draw the molecular orbital diagrams for F2 and F2*. Identify their bond order and magnetic properties. (c) An unstable nucleus exhibit radioactivity. (i) Explain how the number of protons and neutrons in a radioactive nucleus can be used to predict its probable mode decay. (ii) Illustrate your answer in (i) with a schematic graph.arrow_forwardDraw a Lewis electron-dot symbol for (a) Rb; (b) Si; (c) I.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

