
Concept explainers
(a)
Interpretation:
The formal charge on each atom has to be determined and structure of lower energy has to be identified.

Concept Introduction:
The formal charge on each atom in the Lewis structure can be calculated from the equation written as follows:
Here,
Lowest energy structure is the one that has zero or nearly zero formal charge.
(a)
Explanation of Solution
The formal charge on each atom in the Lewis structure can be calculated from the equation as follows:
For structure 1:
Substitute 5 for
Substitute 4 for
Similarly for other Lewis structure, substitute 5 for
Substitute 5 for
Substitute 4 for
Hence formal charge on each structure is illustrated below:

Since the former has less formal charges on maximum number of atoms so I is lower in energy than II therefore I represents structure of lowest energy.
(b)
Interpretation:
The formal charge on each atom has to be determined and structure of lower energy has to be identified.
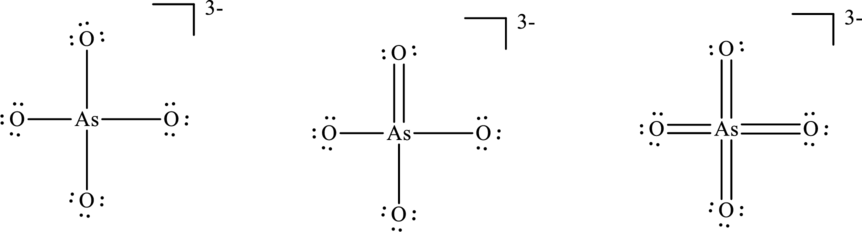
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
The formal charge on each atom in the Lewis structure can be calculated from the equation as follows:
For structure I:
Substitute 6 for
Substitute 6 for
Substitute 5 for
For structure II:
Similarly for second Lewis structure, substitute 6 for
Substitute 6 for
Substitute 5 for
For structure III:
Similarly for third Lewis structure, substitute 6 for
Substitute 5 for
Hence formal charge in the two structures is illustrated below.
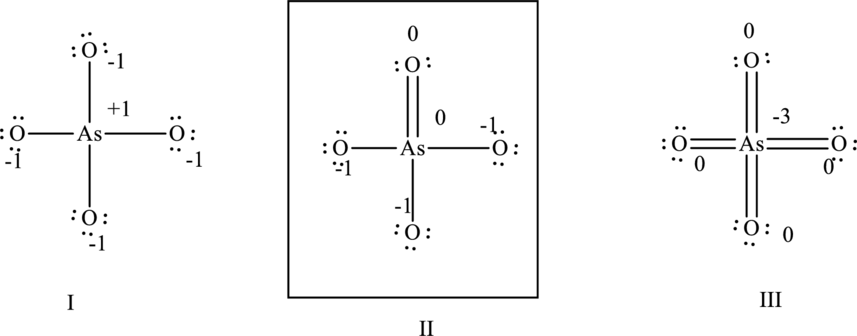
Since the II has a
(b)
Interpretation:
The formal charge on each atom has to be determined and structure of lower energy has to be identified.
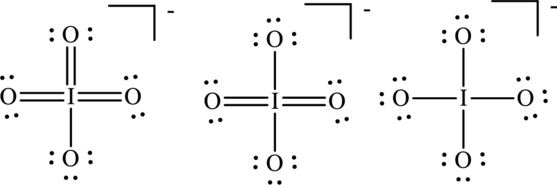
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
The formal charge on each atom in the Lewis structure can be calculated from the equation as follows:
For Lewis structureI, substitute 6 for
Substitute 6 for
Substitute 7 for
For Lewis structure II,
Substitute 6 for
Substitute 6 for
Substitute 7 for
For Lewis structure III, substitute 6 for
Substitute 6 for
Substitute 7 for
Hence formal charge in the three structures is illustrated below:
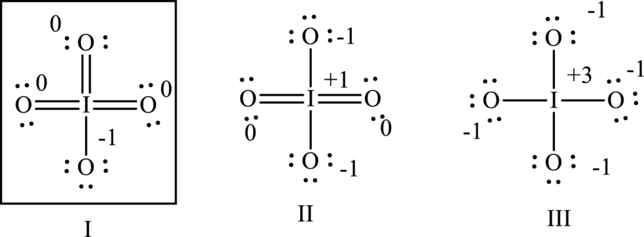
Since I has zero formal charges on central electronegative iodine atom compared to other cases with
Want to see more full solutions like this?
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Assign formal charges to all atoms in the following Lewis diagrams. (a) SO42 (b) S2O32 (c) SbF3 (d) SCNarrow_forwardA complete Lewis structure must show all nonzero formal charges. Complete each of thefollowing Lewis structures by adding any missing formal charges.arrow_forwardWrite the correct Lewis structure and assign a formal charge to each atom in fulminate ion, CNO.arrow_forward
- The molecular ion S3N3 has the cyclic structure All SN bonds are equivalent. (a) Give six equivalent resonance hybrid Lewis diagrams for this molecular ion. (b) Compute the formal charges on all atoms in the molecular ion in each of the six Lewis diagrams. (c) Determine the charge on each atom in the polyatomic ion, assuming that the true distribution of electrons is the average of the six Lewis diagrams arrived at in parts (a) and (b). (d) An advanced calculation suggests that the actual charge resident on each N atom is 0.375 and on each S atom is +0.041 . Show that this result is consistent with the overall +1 charge on the molecular ion.arrow_forwardWrite the Lewis structure and chemical formula of the compound with a molar mass of about 70 g/mol that contains 19.7% nitrogen and 80.3% fluorine by mass, and determine the formal charge of the atoms in this compound.arrow_forwardCarbon monoxide (CO) is an example of an overall neutral molecule (netcharge=0) that hasnon-zero formal charges. Draw a Lewis structure of carbon monoxide (CO).arrow_forward
- Draw resonance structures for each of these ions: NSO− and SNO−. (The atoms are bonded in the order given in each case, that is, S is the central atom in NSO−.) Use formal charges to determine which ion is likely to be more stable. Explain why the two ions cannot be considered resonance structures of each other.arrow_forwardTwo Lewis structures can be written for nitrosyl fluoride, which contains one nitrogen, one oxygen, and one fluorine atom per molecule. Write the two Lewis structures land assign a formal charge to each atom.arrow_forwardThe cyclohexane carboxylate anion has a Lewis structure Pushing a pair of unshared electrons away from the negatively charged oxygen atom and, at the same time, pushing a pair of pi electrons toward the other oxygen will generate a second resonance structure. Thus,arrow_forward
- Chloromethane has the Lewis structure _______________________________ The carbon atom is sharing 4 electron pairs. In each shared pair the carbon atom “owns” 1 electron. The number of electrons that “belong” to carbon is ___. Carbon, being a Group ___ element would have 4 , outer shell electrons in the unbonded, neutral state. Therefore, the carbon atom in chloromethane has a formal charge of zero.arrow_forwardThe Lewis structure of acetone is Circling the carbonyl carbon, i.e., the carbon atom attached to oxygen, and its octet gives Circling the oxygen atom and its octet gives Thus, atoms share electrons in making bonds, and a pair of electrons may be included in the octet of two different atoms. When computing the formal charge on an atom, the number of electrons that belong to that atom is compared with the number of electrons the atom would have in the unbonded and neutral state. If the two numbers are the same, the formal charge on the atom is zero. In a Lewis structure both electrons in an unshared pair belong to the atom, and one of every pair of shared (bonding) electrons belongs to the atom.arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





