
Concept explainers
a)
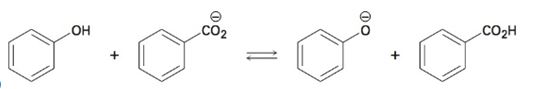
Interpretation:
Using the pKa values the direction in which equilibrium is favored in the reaction given is to be stated.
Concept introduction:
The direction in which equilibrium is favored in the reaction can be ascertained from the pKa values of the substances involved. Lower the pKa value, stronger the acid. Further the relative stabilization by resonance of the conjugate bases produced also has to be considered.
To determine:
The direction in which equilibrium is favored in the reaction using the pKa values given.
b)
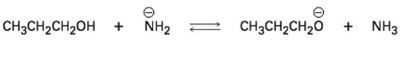
Interpretation:
Using the pKa values the direction in which equilibrium is favored in the reaction given is to be stated.
Concept introduction:
The direction in which equilibrium is favored in the reaction can be ascertained from the pKa values of the substances involved. Lower the pKa value, stronger the acid. Further the relative stabilization by resonance of the conjugate bases produced also has to be considered.
To determine:
The direction in which equilibrium is favored in the reaction using the pKa values.
c)
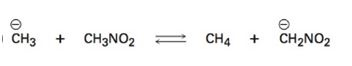
Interpretation:
Using the pKa values the direction in which equilibrium is favored in the reaction given is to be stated.
Concept introduction:
The direction in which equilibrium is favored in the reaction can be ascertained from the pKa values of the substances involved. Lower the pKa value, stronger the acid. Further the relative stabilization by resonance of the conjugate bases produced also has to be considered.
To determine:
The direction in which equilibrium is favored in the reaction using the pKa values.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Organic Chemistry
- During the day, solar radiation is absorbed by NO3(g)NO3(g), resulting in its decomposition. Which of the following best explains whether the equilibrium concentration of N2O5(g)N2O5(g) in the atmosphere in the daytime is different from that in the nighttime, and why?arrow_forwardIllustrate the process of determining the direction of equilibrium using pKa ?arrow_forwardA 1.00 L flask was filled with 2.00 mol gaseous SO2 and 2.00 mol gaseous NO2 and heated. After equilibrium was reached, it was found that 1.30 mol of gaseous NO was present. Assume that the reaction SO2(g) + NO2(g) SO3(g) + NO(g) occurs under these conditions. Calculate the value of the equilibrium constant, K, for this reactionarrow_forward
- What is the equilibrium concentration (in M to three decimal places) of NO2 for the following reaction if the initial concentration of N2O4 was 0.250 M?arrow_forwardThe reaction of bromine gas with chlorine gas, shown below, has a Kc of 7.20 at 200 oC. If a closed vessel was charged with the two reactants, each at an initial concentration of 0.50 M, what would be the equilibrium concentration of Cl2(g)?arrow_forwardThe equilibrium constant, Kc , for the reaction 2 SO2 (g) + O2 (g) → 2 SO3 (g) is 6.90 x 103 . (a) What is Kc for the reaction 2 SO3 (g) → 2 SO2 (g) + O2 (g) (b) What is Kc for the reaction SO2 (g) + 1/2 O2 (g) → SO3 (g)arrow_forward
- Most reaction do not go to completions. Equilibrium is established between the reactants and products. For the reaction of A with B to produce C and D, we can write the chemical equation as: аА + bв + сс + dD where + represents equilibrium between the reactants and products. The extent to which the reaction proceeds to product formation, at a given temperature, is given by the equilibrium constant, Kę. The equilibrium constant is written mathematically (for the above equation) as: [C[[D]* K. = [A]°[B]" where, a, b, c, and d are the stoichiometric coefficients from the balanced chemical equation and the brackets, [ ], indicate molar concentration. 1. For the following reaction: 2A + В + 3с calculate the equilibrium constant, K, if at equilibrium the concentration of A is 0.15M, the concentration of B is 0.20M, and the concentration of C is 0.10M.arrow_forwardConsider the following reaction.HSO4 – (aq) + H2O (l) H3O+ (aq) + SO4 2–(aq) Ka = 0.012 If the initial concentrations are [HSO4–] = 0.50 M, [H3O+] = 0.020 M, and [SO4 2–] = 0.060 M, what are the equilibrium concentrations of the products and reactants?arrow_forwardPhosphorus pentachloride decomposes at high temperatures. PC15 (g) ⇒ PC13 (g) + Cl₂ (g) An equilibrium mixture at some temperature consists of 5.72 g of PC15, 4.86 g of PCl3, and 3.59 g of Cl2 in a 1.00-L flask.arrow_forward
- The reaction 2CO(g) + O2(g) = 2 CO2(g) is known to be exothermic. According to Le Chatelier's principle, how will this reaction's equilibrium be affected if the reaction temperature is increased? (A) The equilibrium constant will decrease and the reaction will shift to the left. (B) The equilibrium constant will be unchanged, but the reaction will shift to the left. (C) The equilibrium constant will increase and the reaction will shift to the right. (D) The equilibrium constant will be unchanged, but the reaction will shift to the right. (E) The equilibrium concentrations will not be affected.arrow_forwardHow is the equilibrium position of this reaction affected by the ADDING of SOa? 2 SO2 (9) + O2 (9) – - 2 Sos (9) + heat How is the equilibrium position of this reaction affected by the REMOVING of 02? 2 SO2 (g) + O2 (9) – - 2 Sos (g) + heatarrow_forwardHydrogen, a potential fuel, is found in great abundance in water. To separate the hydrogen from the oxygen, water must be thermally decomposed. At 1000°C, the equilibrium constant for this reaction is 7.3 x 10-182H2O(g) + heat <-> 2H2(g) + O2(g) a) If the initial concentration of H2O is 0.350 M, what is the equilibrium concentration of H2? b) Suggest three stresses that could be applied to the equilibrium system to produce more hydrogen.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
