
Concept explainers
Skill Building Wavelength, Frequency, and Energy
Return to Figure 3.4, and calculate the amount of energy present in a wave of red light and a wave of blue light. Are they the same amount of energy? Why or why not?
In the not-too-distant past, many people used film exposure to take pictures. A “dark” room was a place where this film was developed into the actual images based on how light exposed the film during the taking of the pictures. These dark rooms used red lights during the time of developing the film because a special dye was added to the film that was not affected by light with a wavelength between 620 and 750 nm. Do you think these rooms could have also used blue lights? Why or why not?
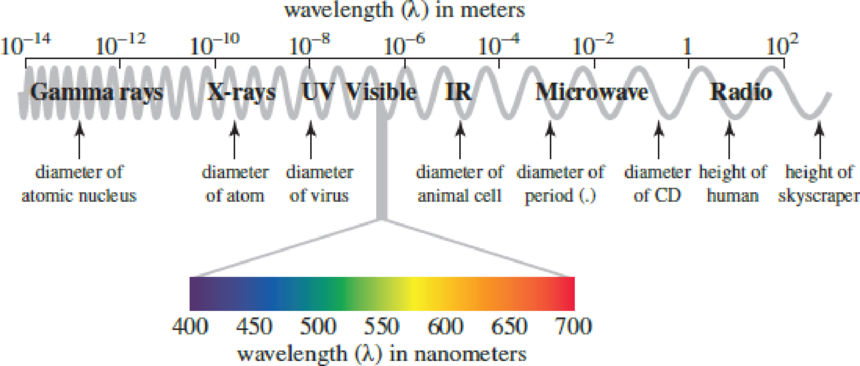
Figure 3.4
The
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Chemistry In Context
Additional Science Textbook Solutions
Organic Chemistry
Essential Organic Chemistry (3rd Edition)
Introductory Chemistry (6th Edition)
Chemistry
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry: Structure and Properties
- The following are representative wavelengths in the infrared, ultraviolet, and x-ray regions of the electromagnetic spectrum, respectively: 1.0 106m, 1.0 108m, and 1.0 1010m. What is the energy of a photon of each radiation? Which has the greatest amount of energy per photon? Which has the least?arrow_forwardThe frequency of the strong red line in the spectrum of potassium is 3.91 1014/s. What is the wavelength of this light in nanometers?arrow_forwardThe element cesium was discovered in 1860 by Robert Bunsen and Gustav Kirchhoff, who found two bright blue lines in the spectrum of a substance isolated from a mineral water. One of the spectral lines of cesium has a wavelength of 456 nm. What is its frequency?arrow_forward
- Use the building-up principle to obtain the electron configuration for the ground state of the manganese atom (Z = 25).arrow_forwardAn atom has a line spectrum consisting of a red line and a blue line. Assume that each line corresponds to a transition between two adjacent energy levels. Sketch an energy-level diagram with three energy levels that might explain this line spectrum, indicating the transitions on this diagram. Consider the transition from the highest energy level on this diagram to the lowest energy level. How would you describe the color or region of the spectrum corresponding to this transition?arrow_forwardINTERACTIVE EXAMPLE Energy Quantization in Hydrogen Complete the statement: When a hydrogen electron is excited from level n 3 to level n 5, a photon of wavelength m is absorbed. HOW DO WE GET THERE? Calculate E, for the n = 3 energy level. J Check Next (2 of 5) Submit Answer Try Another Version 1 item attempt remainingarrow_forward
- DETAILS MY NOTES Use the Rydberg equation to calculate the wavelength (in Å) of the photon absorbed when a hydrogen atom undergoes a transition from n = 1 to n = 4. (Report your answer to at least 3 significant figures.) Submit Answerarrow_forwardEXERCISE 1 Calculate the rotational energies of the first four energy levels of the HI molecule knowing that the bond length is r = 160 pm and that rotation in all three dimensions about the center of mass is allowed. The atomic masses of H = 1.00794 amu and I = 126.9045 amu are given.arrow_forwardCrop the photo 2 Using the table above, calculate the frequency of red light. A 4.6x 1014HZ B 3.0 x 10 Hz C 1.3 x 10 Hz D 9.8x 10 Hz 3 Using the table above, calculate the wavelength of blue light. A 3.9 x 10-3 m B 22x 10-7 m C 3,0 x 10- m D 4.5x 10- m 4 How much energy in joules is found in a photon o yellow liohtwith wavelength 5.9 × 10-7 m? A 1.3 Crop the ONE problem you B 3.9x 10- need help with C 3.4 107 D 3.4 x 1014T Which element has the noble gas notationarrow_forward
- CHEMWORK Check all the boxes that are true. (Select all that apply.) O The third energy level has three sublevels - the s, p, and d sublevels. In going from the 2s' state to the 1s' state, a hydrogen atom would emit a photon of light. O The hydrogen atom has quantized energy levels. In the buildup of atoms, the 3d orbital fills before the 4s orbital. Blue light is less energetic than red light. Submitarrow_forwardTopic: Science Technology for the 21st Century 1. What is the key to unlock to door to prosperity in the Philippines? 2. Enumerate 10 advancements in technology that make our life, business and agriculture easier. 3. What is a bad effect of synthetic fertilizer? 4. How is laser used in medicine? What is the meaning of the acronym LASER? 5. How does Photon work in medicine? 6. List 6 uses of LASER. 7. What are fiber optics? 8. How is this used in medicine? 9. Aside from medicine, where else are fiber optics used? 10. Where are the following optics used? a. Side Emitting Fiber Optics b. Tip Illuminated Fiber Optics c. Fiber Optic Light Pipes 11. How does light travel? 12. How does a prism split white light into a spectrum? 13. How can we produce Magenta, cyan, and yellow? 14. How does a pinhole camera work?arrow_forwardName the scientist who demonstrated photoelectric effect experiment.arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

