
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 4.40SP
Label each hydrogen atom in the following compounds as primary (1°), secondary (2°), or tertiary (3°).
- a. CH3CH2CH(CH3)2
- b. (CH3)3CCH2CH3
- c. (CH3)2CHCH(CH3)CH2CH3
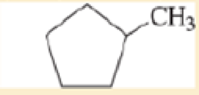
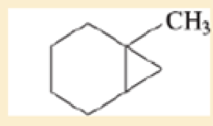
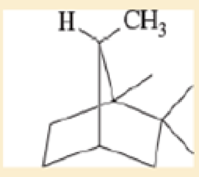
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
4. Use the IUPAC Nomenclature System to name each of the following compounds:
a.
b.
CH,CCH₂CH,
0
HCCHCH₂CH₂
CI O
-C-C-CH₂
Br
CHỊCH,CH,CH,
CH,CCH₂CH₂CH
CH,
CI
CH₂CHCH₂CH
Br
CH, Ô
CH,CCH.CCH.CH.CH,
OH
0
CH₂
CH,CCH₂CH.CH,CHCH,C-H
H,CHCH.CH
CH₂
Part II (70 points)
Directions: Answer each of the questions that follow. Please read the
directions for each question carefully. When structures are required please
draw them Clearly and Neatly. Make sure questions are numbered
clearly as well.
1. (12 points; 2 points each) Give a correct name for each of the following compounds:
CH3
A) CH—CH,C—CHICHCHCH, CH
OH
alf
B) CH-CH₂-
CH₂
-CH₂-CH₂
How many asymmetric carbons are there in the compound below?
CH3
H₂C-N.
CH, OH
D
OA. TWO
B.One
OC. Four
OD. Three
O E. Five
-CEC-CH3
Chapter 4 Solutions
Organic Chemistry (9th Edition)
Ch. 4.3A - Draw Lewis structures for the following free...Ch. 4.3B - a. Write the propagation steps leading to the...Ch. 4.3C - Prob. 4.3PCh. 4.3C - Prob. 4.4PCh. 4.4 - The following reaction has a value of G =...Ch. 4.4 - Under base-catalyzed conditions two molecules of...Ch. 4.5B - When ethene is mixed with hydrogen in the presence...Ch. 4.5B - For each reaction, estimate whether S for the...Ch. 4.7 - a. Propose a mechanism for the free radical...Ch. 4.7 - a. Using bond-dissociation enthalpies from...
Ch. 4.8 - The reaction of tert-butyl chloride with methanol...Ch. 4.8 - Under certain conditions, the bromination of...Ch. 4.8 - When a small piece of plat num is added to a...Ch. 4.10 - Prob. 4.14PCh. 4.10 - Prob. 4.15PCh. 4.12 - The bromination of methane proceeds through the...Ch. 4.12 - a. Using me BDEs in Table4-2 (page 167 ), compute...Ch. 4.13A - What would be the product ratio in the...Ch. 4.13A - Classify each hydrogen atom in the following...Ch. 4.13B - Use the bond-dissociation enthalpies in Tabte4-2...Ch. 4.13B - Prob. 4.21PCh. 4.13B - Prob. 4.22PCh. 4.14 - a. Compute the heats of reaction for abstraction...Ch. 4.14 - 2,3-Dimethylbutane reacts with bromine in the...Ch. 4.14 - Prob. 4.25PCh. 4.15 - Prob. 4.26PCh. 4.15 - Prob. 4.27PCh. 4.16A - Prob. 4.28PCh. 4.16A - Prob. 4.29PCh. 4.16B - Prob. 4.30PCh. 4.16C - Prob. 4.31PCh. 4.16C - Acetonitrile (CH3C N) is deprotonated by very...Ch. 4.16D - Prob. 4.33PCh. 4 - The following reaction is a common synthesis used...Ch. 4 - Consider the following reaction-energy diagram. a....Ch. 4 - Draw a reaction-energy diagram for a one-step...Ch. 4 - Draw a reaction-energy diagram for a two-step...Ch. 4 - Prob. 4.38SPCh. 4 - Treatment of tert-butyl alcohol with concentrated...Ch. 4 - Label each hydrogen atom in the following...Ch. 4 - Prob. 4.41SPCh. 4 - Prob. 4.42SPCh. 4 - Prob. 4.43SPCh. 4 - Prob. 4.44SPCh. 4 - Prob. 4.45SPCh. 4 - Prob. 4.46SPCh. 4 - For each compound, predict the major product of...Ch. 4 - When exactly 1 mole of methane is mixed with...Ch. 4 - Prob. 4.49SPCh. 4 - Prob. 4.50SPCh. 4 - Prob. 4.51SPCh. 4 - When dichloromethane is treated with strong NaOH,...Ch. 4 - Prob. 4.53SPCh. 4 - When a small amount of iodine is added to a...Ch. 4 - Prob. 4.55SPCh. 4 - When healthy, Earths stratosphere contains a low...Ch. 4 - Prob. 4.57SPCh. 4 - lodination of alkanes using iodine (I2) is usually...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following structures have the correct IUPAC names? CI 1 111 H CH3 (R)-2-chloropentane OCH 3 H CH CH₂CH₂CH3 Br-C- RS-1,2-dibromo-1,2-dimethoxyethane O a. O b. I and III O c. I and IV O d. II and III III and IV C-Br OCH 3 || D IV (S)-4-fluoro-4-methylcyclohexene CI CH3 CH3 "F CF CH₂CH3 (S)-2-chloro-2-fluoro butanearrow_forward1. Give the IUPAC name for the following compounds: a. C. e. OH O || b. CH3–CH2–C–0–CH2–CH2–CH3 CH3–CH,—NH, Ο CH3 d. CH3-N-CH₂-CH3 Η | || CH3-CH₂-CH₂-C-N-CH₂-CH3arrow_forwardWhich of the following compounds is chrally? CH3 CH, CH H. H3C. "CH;CH3 CH, Br H. H Br 1 3. One answer must be chosen: only 2 -11 O 3 -1 1,2 O only a1 O only 3 -1 2 Oarrow_forward
- Select the single best answer. Classify the pair of compounds as constitutional isomers or identical molecules. H. CH3 H. and CH3 CH3 CH(CH3)2 identical molecules constitutional isomers エarrow_forward1. Identify the circled functional group(s) for each molecule. COM OH a. b. C. d. CH3CHCH3 O NH,CH,CH,CH,NHCH3 H₂N C C=CH₂ H₂ C H 0- -CH₂CHCH NHCH(CH3)2 OHarrow_forwardIdentify the best reagents to complete the following reaction. CI N.arrow_forward
- Circle and the functional group (5) in each compound. name キ の. CHz CH3 NH C - Br b.CH3 CH= CH CHz- CH3 -CH-arrow_forward1 a Name each of the following compounds: D H3C- A CH3 CH3 HạC—CH—CH, NH, C−CH,NH, CH 3 E B H3C- CH3 HỌC—-CH-CHNH, CH₂ CH3 H₂C-C=C- -NH₂ CH3 F Oe -NH₂ -CHz—CHz—NHzarrow_forwardWhat is each compound’s systematic name? 1. (CH3)3CCH2CH2CH2CH(CH3)2 2. BrCH2CH2CH2CH2CH2NHCH2CH3 3.(CH3CH2)4C 4.CH3OCH2CH2CH2OCH3arrow_forward
- Which of the following is not another representation for 2-methylbutane? H H₂CH₂C+CH₂ CH₂ 1 Multiple Choice CH₂CH₂CH(CH₂)2 111 HH H-C-C-H H₂C- CH₂ CH₂ IVarrow_forwardА H. OH C CH,CH3 D E NH CH2CH3 CH2CH,arrow_forward10. Assigh E or Z configuration for each alkene shown below. CEN H3CH 2C. CH2NH2 H3C HOarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY