
Concept explainers
a)
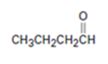
Interpretation:
The product obtained in the aldol reaction of butanal is to be given.
Concept introduction:
To give:
The product obtained in the aldol reaction of butanal.
b)
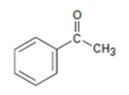
Interpretation:
The product obtained in the aldol reaction of acetophenone is to be given.
Concept introduction:
Aldehydes and ketones with α-hydrogen undergo a base catalyzed carbonyl condensation reaction in aldol condensation. In this reaction two molecules of the reactant combine by forming a bond between α-carbon of one molecule and the carbonyl carbon of the second molecule. The product is a β-hydroxyaldehyde or ketone.
To give:
The product obtained in the aldol reaction of acetophenone.
c)
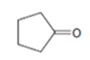
Interpretation:
The product obtained in the aldol reaction of cyclopentanone is to be given.
Concept introduction:
Aldehydes and ketones with α-hydrogen undergo a base catalyzed carbonyl condensation reaction in aldol condensation. In this reaction two molecules of the reactant combine by forming a bond between α-carbon of one molecule and the carbonyl carbon of the second molecule. The product is a β-hydroxyaldehyde or ketone.
To give:
The product obtained in the aldol reaction of cyclopentanone.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- Describe how the following compound can be prepared using an aldol addition in the first step of the synthesis:arrow_forwardDescribe how the following compounds can be prepared using an aldol addition in the first step of the synthesis:arrow_forwardThe Stork reaction is a condensation reaction between an enamine donor and an α,β-unsaturated carbonyl acceptor. The overall reaction consists of a three-step sequence of formation of an enamine from a ketone, Michael addition to an α,β-unsaturated carbonyl compound, and hydrolysis of the enamine in dilute acid to regenerate the ketone. Consider the Stork reaction between cyclohexanone and propenal Draw the structure of the product of the enamine formed between cyclohexanone and dimethylamine. - Michael addition to an α,β-unsaturated carbonyl compound, and - hydrolysis of the enamine in dilute acid to regenerate the ketone.arrow_forward
- Identify the major aldol condensation product obtained when the following compound is heated in the presence of aqueous sodium hydroxide.arrow_forwardPredict the four possible Aldol condensation products and their dehydration products. KOH, H,O, Aarrow_forwardPredict the major product in each of the following reactions:arrow_forward
- The ketone whose 1H NMR spectrum is shown here was obtained as the product of an acetoacetic ester synthesis. What alkyl halide was used in thesynthesis?arrow_forwardPredict the outcome of the intramolecular aldol condensation of the following compounds:arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forward
- Predict the major products of the following base-catalyzed aldol condensations withdehydration.(a) benzophenone (PhCOPh) + propionaldehyde(b) 2,2@dimethylpropanal + acetophenonearrow_forwardThe ketone whose 1H NMR spectrum is shown here was obtained as the product of an acetoacetic ester synthesis. What alkyl halide was used in the synthesis?arrow_forwardPredict the products of aldol condensation, followed by dehydration, of the followingketones and aldehydes. c) cyclohexanonearrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

