
Concept explainers
a)
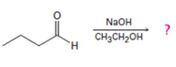
Interpretation:
The dehydration product formed in the addition reaction of butanal along with the mechanism of its formation is to be predicted.
Concept introduction:
The β- hydroxyl aldehyde or ketone obtained loses a molecule of water upon heating to give an α, β- unsaturated aldehyde or ketone.
To predict:
The dehydration product formed in the addition reaction of butanal along with the mechanism of its formation.
Answer to Problem 28MP
The dehydration product formed in the addition reaction of butanal is 2-ethyl-2-hexenal.
The mechanism of its formation is
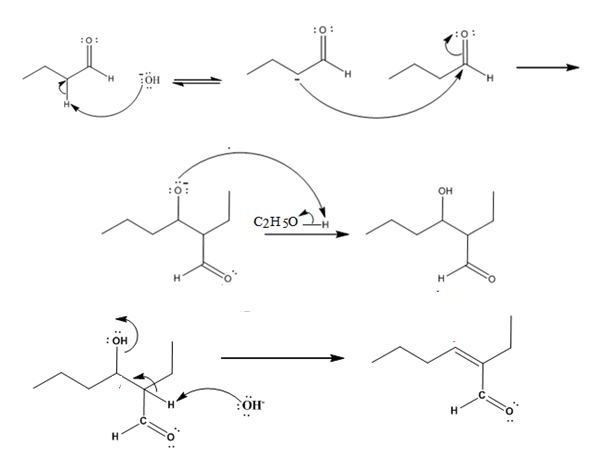
Explanation of Solution
In the first step the base picks up an acidic proton from the diketone to produce the enolate anion. In the next step the nucleophilic enolate anion attacks the electrophilic carbon of the other carbonyl group in the same molecule to give an alkoxide. In the next step the alkoxide intermediate is protonated to yield the β- hydroxyl aldehyde. Removal of a proton by the base from the hydroxy aldehyde leads to the formation of α, β- unsaturated aldehyde.
The dehydration product formed in the addition reaction of butanal is 2-ethyl-2-hexenal.
The mechanism of its formation is

b)
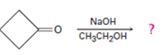
Interpretation:
The dehydration product formed in the addition reaction given along with the mechanism of its formation is to be predicted.
Concept introduction:
Aldehydes and ketones that have α- hydrogen atom undergo aldol condensation to yield a β- hydroxyl aldehyde or ketone as the product. The reaction occurs in three steps i) Abstraction of α- hydrogen by a base to yield an enolate anion ii) Attack of the anion on the carbonyl carbon of another molecule iii) Protonation of the alkoxide intermediate.
The β- hydroxyl aldehyde or ketone obtained on heating loses a water molecule to yield an α, β- unsaturated aldehyde or ketone.
To identify:
The dehydration product formed in the addition reaction of cyclobutanone along with the mechanism of its formation.
Answer to Problem 28MP
The dehydration product formed in the addition reaction of cyclobutanone is
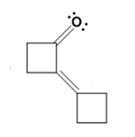
The mechanism of its formation is
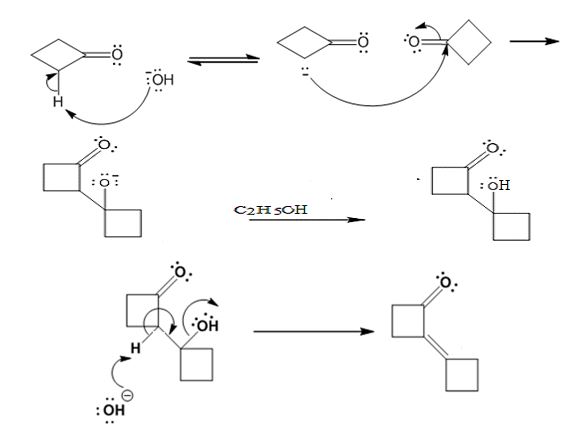
Explanation of Solution
In the first step the base picks up a proton from the α-carbon of one cyclobutanone molecule to produce the enolate anion. In the next step the nucleophilic enolate anion attacks the electrophilic carbonyl carbon of another cyclobutanoe molecule to give an alkoxide. In the next step the alkoxide intermediate is protonated to yield a β- hydroxyketone. In the final step the base picks up a proton from the hydroxyl group that leads to the formation of α, β- unsaturated ketone.
The dehydration product formed in the addition reaction of cyclobutanone is

The mechanism of its formation is

c)
Interpretation:
The dehydration product formed in the addition reaction given along with the mechanism of its formation is to be predicted.
Concept introduction:
The reaction given is a mixed aldol reaction. Aldehydes and ketones that have α- hydrogen atom undergo aldol condensation to yield a β- hydroxyl aldehyde or ketone as the product. The reaction occurs in three steps i) Abstraction of α- hydrogen by a base to yield an enolate anion ii) Attack of the anion on the carbonyl carbon of another molecule iii) Protonation of the alkoxide intermediate.
The β- hydroxyl aldehyde or ketone loses a molecule of water when heated in the presence of a base to yield α, β- unsaturated aldehyde or ketone.
To identify:
The dehydration product formed in the addition reaction given along with the mechanism of its formation.
Answer to Problem 28MP
The dehydration product formed in the addition reaction given is
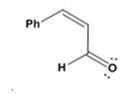
The mechanism of its formation is
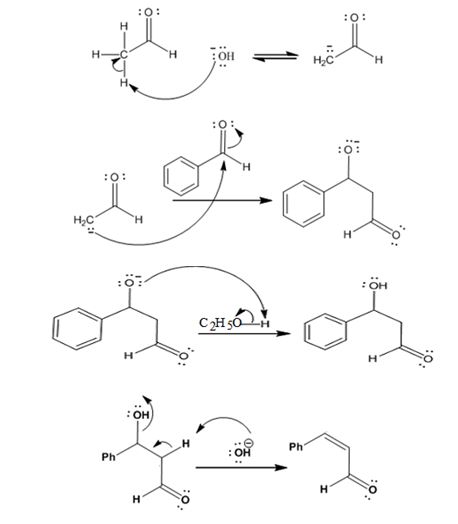
Explanation of Solution
In the first step the base picks up a proton from the α-carbon of acetaldehyde as it has α- hydrogen atoms to produce the enolate anion. In the next step the nucleophilic enolate anion attacks the electrophilic carbonyl carbon of benzaldehyde to give an alkoxide. In the next step the alkoxide intermediate is protonated to yield a β- hydroxyaldehyde. In the final step the base picks up a proton from the hydroxyl group that leads to the formation of α, β- unsaturated aldehyde.
The dehydration product formed in the addition reaction given is

The mechanism of its formation is

d)
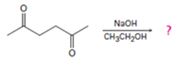
Interpretation:
The product formed in the addition reaction given along with the mechanism of its formation is to be predicted.
Concept introduction:
In intramolecular aldol reactions dicarbonyl compounds such as diketones react with a base to yield a cyclic enone as the products. The reaction occurs in four steps i) Abstraction of α-hydrogen by a base to yield an enolate anion ii) Attack of the anion on the carbonyl carbon in another molecule iii) Protonation of the alkoxide intermediate. iv) Loss of water from the keto alcohol upon heating.
To identify:
The product formed in the addition reaction given along with the mechanism of its formation.
Answer to Problem 28MP
The product formed in the addition reaction given is
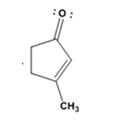
The mechanism of its formation is
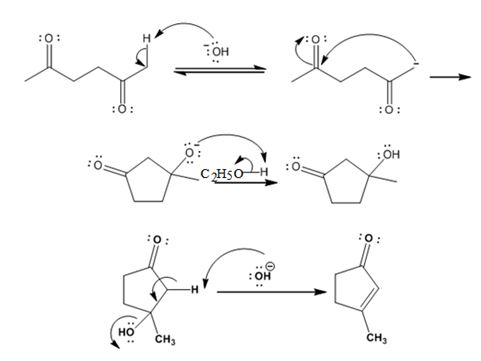
Explanation of Solution
In the first step the base picks up an acidic proton from the diketone to produce the enolate anion. In the next step the nucleophilic enolate anion attacks the electrophilic carbon of the other carbonyl group in the same molecule to give an alkoxide. In the next step the alkoxide intermediate is protonated to yield a hydroxyl ketone. In the final step the base picks up a proton from the hydroxyl group that leads to the formation of α, β- unsaturated aldehyde.
The product formed in the addition reaction given is

The mechanism of its formation is

Want to see more full solutions like this?
Chapter 23 Solutions
Organic Chemistry
- b) Refer to the following equation to answer Q3b (i), (ii) and (iii). CH3 H,SO, Н—с—он C-CH3 ? + H2O Но- ČH3 (i) Determine the product of the above reaction. (ii) Name the above reaction. (iii) Propose the mechanism for the above reaction.arrow_forwardN 3 5 Predicate the products of each reaction: Aldehydes + Ketones CH₂-C-OCH, CH;CH + HC=N =O + H₂NCH₂ OH O + H₂O H H₂SO4 HgSO4 2 H₂ Ni NaCN + NaBH₂ H+ Zn (Hg), HCIarrow_forward21) Propose a mechanism for the following reaction. OH H20, H*arrow_forward
- Reaction with which of the following compounds in an acetylide reaction would lengthen the carbon chain by one carbon and add a primary alcohol? O ethanal O formaldehyde О ерохide O acetone O carbonic acid QUESTION 4 The reagent/reaction of choice for this reduction is || HC-CH=CH-CH2CH2CH2 CO2H CH2OH-CH=CH-CH2CH2CH2 CO2H O lithium aluminum hydride O potassium permanganate O sodium borohydride O Grignard reaction O SN2 reactionarrow_forwardExplain mechanism - Base-Catalyzed Addition of H2O to a Carbonyl Group ?arrow_forwardHN- OH GH Provide synthesis of the product and show the mechanism. please consider the stereochemistry of the product.arrow_forward
- Amantadine is effective in preventing infections caused by the influenza A virus and in treating established illnesses. It is thought to block a late stage in the assembly of the virus. Amantadine is synthesized by treating 1-bromoadamantane with acetonitrile in sulfuric acid to give N-adamantylacetamide, which is then converted to amantadine. CH,C=N in H,SO, Br NHCCH3 NH2 1-Bromoadamantane Amantadine (a) Propose a mechanism for the transformation in Step 1. (b) Describe experimental conditions to bring about Step 2.arrow_forwardGive a mechanism that accounts for the organic products of this reaction. Your mechanism must include a transition state that accounts for the stereochemistry of the product and/or reactant. Redraw the reagents as needed. CI OH NaOH, DMSO + NaClarrow_forward4) Propose a synthetic pathway to prepare 2-methyl-propan-2-ol from propane. You're allowed to use any reagents you like but all the carbon atoms in your product should come from the propane. он It must be propanone otherwise the conversion would be too long.arrow_forward
- When a methyl ester is hydrolyzed under acidic conditions in H,180, the 180 isotope ends up in the carboxylic acid. When a tert-butyl ester is hydrolyzed under the same conditions, the labeled oxygen ends up in the alcohol product. (a) Propose mechanisms to account for these observations. (b) Explain why each ester undergoes the respective mechanism. CH3 + Но-СНЗ 18 + H180 H ОНarrow_forwardWhat products are formed when the following molecule reacts with catalytic H2SO4, H2O, and heat? Please show the mechanism for the reaction.arrow_forwardShow the complete mechanism using Curved Arrow Formalism for the reaction scheme below in acidic conditions. Name the functional group generated and tell how many peaks would be visualized in the 13C NMR spectrum for the starting material and product generated. HO O OH + H₂Oarrow_forward
