
a)
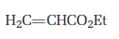
Interpretation:
The product that will be produced when the enamine obtained by reacting cyclopentanone and pyrrolidone with α, β- unsaturated acceptor, ethyl acrylate, is to be identified.
Concept introduction:
The enamine obtained by reacting a
To identify:
The product that will be produced when the enamine obtained by reacting cyclopentanone and pyrrolidone with α, β- unsaturated, ethyl acrylate, is hydrolyzed.
b)
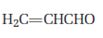
Interpretation:
The product that will be produced when the enamine obtained by reacting cyclopentanone and pyrrolidone with α, β- unsaturated acceptor, acrolein is to be identified.
Concept introduction:
The enamine obtained by reacting a ketone with cyclic amine reacts with α, β- unsaturated acceptor, acrolein, to yield a substituted enamine as the intermediate which upon hydrolysis gives the products.
To identify:
The product that will be produced by the hydrolysis of the enamine obtained by reacting cyclopentanone and pyrrolidone with α, β- unsaturated acceptor, acroline.
c)
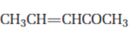
Interpretation:
The product that will be produced when the enamine obtained by reacting cyclopentanone and pyrrolidone with α, β- unsaturated acceptor, pent-3-ene-2-one, is to be identified.
Concept introduction:
The enamine obtained by reacting a ketone with cyclic amine reacts with an α, β- unsaturated acceptor to yield a substituted enamine as the intermediate which upon hydrolysis gives the products.
To identify:
The product that will be produced by the hydrolysis of the enamine obtained by reacting cyclopentanone and pyrrolidone with α, β- unsaturated acceptor, pent-3-ene-2-one.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Organic Chemistry
- How would you carry out the following reactions? More than one step may be required. (a) 3-Hexyne →→ 3-Hexanone (b) Benzene →→ m-Bromoacetophenone (c) Bromobenzene →→ Acetophenone (d) 1-Methylcyclohexene →→ 2-Methylcyclohexanonearrow_forwardReaction of p-nitroaniline with sodium nitrite and hydrochloric acid at 0°C, followed by treatment with N,N-diethylaniline.arrow_forwardThe reaction of cyclohexanone and N,N-dimethylamine with sodiumtriacetoxyborohydride (NaBH(OAc)3) in methanol with acetic acid (AcOH) affords N,Ndimethylcyclohexanamine in high yield. However, if anhydrous HCl is used the reaction yield of N,Ndimethylcyclohexanamine is low and a side reaction occurs to produce alcohol. Draw a reaction mechanism for the reductive amination sequence, highlighting tautomers present in the reaction.arrow_forward
- Draw the main organic product formed in each of the following reactions: i NaOH (a) (b) O + H₂O OCH 3 OCH 3 + NH3 + CH3CH₂OH OCH3 HCI attach to this assignment.arrow_forward17-48 How would you prepare the following compounds from More than one step may be required. 1-phenylethanol? (a) Acetophenone (PhCOCH3) (b) Benzyl alcohol (c) m-Bromobenzoic acid (d) 2-Phenyl-2-propanol 17-49 How would you prepare the following substances from cyclopentanol? More than one step may be required. (a) Cyclopentanone (b) Cyclopentene (c) 1-Methylcyclopentanol (d) trans-2-Methylcyclopentanol 17-50 What products would you expect to obtain from reaction of 1-methyl- cyclohexanol with the following reagents? (a) HBr (b) NaH (c) H₂SO4 (d) Na₂Cr₂O7arrow_forwardTreating trimethylamine with 2-chloroethyl acetate gives acetylcholine as its chloride. Acetylcholine is a neurotransmitter. Me;N + CH,COCH,CH,CI C,H1,CINO, Acetylcholine chloride Propose a structural formula for this quaternary ammonium salt and a mechanism for its formation.arrow_forward
- Which of the following will more likely undergo an E1cB reaction with NaOH? (A) 4-hydroxybutan-2-one B) 2-chloropentanal (c) butanoic acid D butanoyl chloridearrow_forwardCompound H (C8H6O3) gives a precipitate when treated with hydroxylamine in aqueous ethanol and a silver mirror when treated with Tollens solution. Following is its 1H-NMR spectrum. Deduce the structure of compound H.arrow_forwardTreatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.arrow_forward
- Dihydropyran is synthesized by treating tetrahydrofurfuryl alcohol with an arenesulfonic acid, ArSO3H. Propose a mechanism for this conversion.arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardA step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


