
Pushing Electrons
4th Edition
ISBN: 9781133951889
Author: Weeks, Daniel P.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 3, Problem 32EQ
Interpretation Introduction
Interpretation:
The products of protonation on the
Concept Introduction:
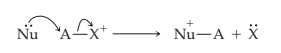
The simultaneous making and breaking of sigma bonds occurs also between a neutral nucleophile or base with pushable electrons and a positively charged species that can act as a receptor.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What explains why many aldehydes and ketones can undergo self-condensation reactions in
basic conditions?
The alpha carbon can lose a proton and act like a nucleophile and the carbonyl
carbon is an electrophile.
The alpha carbon can gain a proton and act like an electrophile and the carbonyl
carbon is a nucleophile.
The oxygen of the carbonyl group can attack the carbon of the carbonyl group.
Only esters can undergo self-condensation reactions.
Write the product formed as a result of the following reaction with its mechanism.
Hydration of aldehydes and ketones can be catalyzed by acid
or base. Bases catalyze hydration by:
protonating the carbonyl oxygen
making the carbonyl group more electrophilic
employing hydroxide ion, which is a better nucleophile than water
making the carbonyl group less electrophilic
shifting the equilibrium position of the reaction to favor products
Chapter 3 Solutions
Pushing Electrons
Ch. 3 - Prob. 1EQCh. 3 - Prob. 2EQCh. 3 - Prob. 3EQCh. 3 - Prob. 4EQCh. 3 - Prob. 5EQCh. 3 - Prob. 6EQCh. 3 - Here are some exercises in sigma bond breaking....Ch. 3 - Prob. 8EQCh. 3 - Prob. 9EQCh. 3 - Prob. 10EQ
Ch. 3 - Prob. 11EQCh. 3 - Prob. 12EQCh. 3 - Prob. 13EQCh. 3 - Prob. 14EQCh. 3 - Prob. 15EQCh. 3 - Prob. 16EQCh. 3 - Prob. 17EQCh. 3 - Prob. 18EQCh. 3 - Prob. 19EQCh. 3 - Prob. 20EQCh. 3 - Prob. 21EQCh. 3 - Prob. 22EQCh. 3 - Prob. 23EQCh. 3 - Prob. 24EQCh. 3 - Prob. 25EQCh. 3 - Prob. 26EQCh. 3 - Prob. 27EQCh. 3 - Prob. 28EQCh. 3 - Prob. 29EQCh. 3 - Prob. 30EQCh. 3 - The reaction just described is reversible....Ch. 3 - Prob. 32EQCh. 3 - Prob. 59EQ
Knowledge Booster
Similar questions
- Consider the mechanism for the given nucleophilic substitution reaction. 2CH CH OHarrow_forwardAn electron-deficient carbon atom reacts with a nucleophile, symbolized as: Nu−. Define this ?arrow_forwardWhat is the slow, rate-determining step, in the acid-catalyzed dehydration of 2- butanol? Loss of a b-hydrogen from the carbocation to form an alkene. Protonation of the alcohol to form an oxonium ion. Loss of water from the oxonium ion to form a carbocation. The simultaneous loss of a B-hydrogen and water from the oxonium ion.arrow_forward
- The following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place.arrow_forwardWhat explains why many aldehydes and ketones can undergo self- condensation reactions in basic conditions? The alpha carbon can lose a proton and act like a nucleophile and the carbonyl carbon a an electrophile O The alpha carbon can gain a proton and act like an electrophile and the carbonyl carbon is a nucleophile O The oxygen of the carbonyl group can attack the carbon of the carbonyl group Only esters can undergo self-condensation reactionsarrow_forwardBy taking into account electronegativity differences, draw the products formed by heterolysis of the carbon–heteroatom bond in each molecule. Classify the organic reactive intermediate as a carbocation or a carbanion.arrow_forward
- write mechanism for the reaction ?arrow_forwardConsider the reaction mechanism for the formation of bromoethane from an alkene and a bromide acid. Draw the structure of the carbonium formed in this reaction.arrow_forwardComplete the missing components of the products in the acid hydrolysis of methyl butanoate below. For the first product fill in the remaining part of the acid formula, and for the second fill in the remaining part of the alcohol formula. Metylbutanoate+H2O--H+-COOH+COOHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
