
Concept explainers
Interpretation:
The labeled bonds in the given compound are to be arranged in the order of increasing bond lengths.
Concept introduction:
In hybridization, one
Answer to Problem 4.1P
The increasing order of bond-lengths in the given compound is
Explanation of Solution
The given compound is,
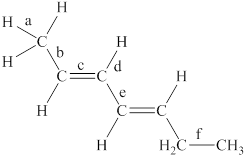
Figure 1
The bond with higher percentage of s-character has electron density closer to the nucleus and thus, has shorter bond length. The order of percentage
• Bond ‘a’ is present between the
• Bond ‘c’ is present between the two
• Due to the conjugation between two pi bonds, the bond length of ‘e’ is in between the carbon-carbon single bond and carbon-carbon double bond.
• Bond ‘b’ is present between one
• The
From the above points, it is concluded that the order of bond-lengths in the given compound is
The increasing order of bond-lengths in the given compound is
Want to see more full solutions like this?
Chapter 4 Solutions
Organic Chemistry
- Also can you provide an explanation along with the drawing as to how you come up with the answer please.arrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided resonance structures, draw the curved electron- pushing arrows to show the interconversion between resonance hybrid contributors. Be sure to account for all bond- breaking and bond-making stepsarrow_forwardPlease respond to the question in the image below.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
