
Concept explainers
(a)
Interpretation:
The
Concept introduction:
Answer to Problem 4.47AP
The configuration assigned to the given structure is
Explanation of Solution
The
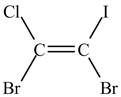
Figure 1
In this case, bromide ion gets priority over chloride ion and iodide ion gets priority over chloride ion. The structure with higher priority order is written as shown below.
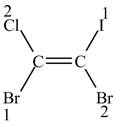
Figure 2
The higher priority groups are present on opposite sides of the double bond. As a result,
The configuration of the given structure is
(b)
Interpretation:
The
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
Answer to Problem 4.47AP
The configuration assigned to the given structure is
Explanation of Solution
The
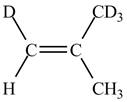
Figure 3
In this case, the
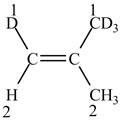
Figure 4
The higher priority atoms are present on the same side of the double bond. As a result,
The configuration of the given structure is
(c)
Interpretation:
The
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
Answer to Problem 4.47AP
The configuration assigned to the given structure is
Explanation of Solution
The
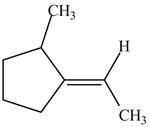
Figure 5
In this case, right side of double bond contains
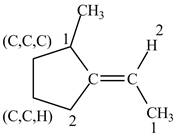
Figure 6
One carbon atom is attached to three carbon atoms and other attached to two carbon atoms. Therefore, the carbon atom which is attached to three carbon atoms gets higher priority. As a result, higher priority atoms are present on opposite sides of the double bond. The structure is assigned
The configuration of the given structure is
(d)
Interpretation:
The
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
Answer to Problem 4.47AP
The configuration assigned to the given structure is
Explanation of Solution
The
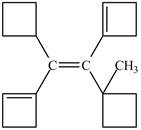
Figure 7
In this case, left and right side of the double bond contains a carbon atoms cyclic ring whose priority order is identified on the basis of higher priority order atoms attached to its carbon atom which is marked as shown below.
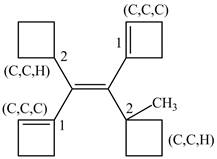
Figure 8
On both sides, One carbon atom is attached to three carbon atoms and other attached to two carbon atoms. Therefore, the carbon atom which is attached to three carbon atoms gets higher priority. Along with double bond get higher priority over the single bond. As a result, higher priority atoms are present on opposite sides of the double bond. The structure is assigned
The configuration of the given structure is
Want to see more full solutions like this?
Chapter 4 Solutions
Organic Chemistry
- What will be the color of the flame and the amount of soot if the following are ignited:(a) Hexane (b)Heptane(c) Cyclohexane (d) Cyclohexene (e) Benzene (f) Toluenearrow_forwardAcid-catalyzed dehydration of 2,2-dimethyl-1-hexanol gave a number of isomeric alkenes including 2-methyl-2-heptene as shown in the following equation.(a) Write a stepwise mechanism for the formation of 2-methyl-2-heptene, using curved arrows to show the flow of electrons. (b) What other alkenes do you think are formed in this reaction?arrow_forward(a) One test for the presence of an alkene is to add a smallamount of bromine, which is a red-brown liquid, and lookfor the disappearance of the red-brown color. This test doesnot work for detecting the presence of an aromatic hydrocarbon.Explain. (b) Write a series of reactions leading topara-bromoethylbenzene, beginning with benzene andusing other reagents as needed. What isomeric side productsmight also be formed?arrow_forward
- H H Write the structures of the alkenes that would yield the following carbonyl compounds PRACTICE PROBLEM 8.22 when treated with ozone and then with dimethyl sulfide. (a) and (c) EO and `H oalio (2 mol is produced from 1 mol of alkene) (b) H.arrow_forwardGiven each of the IUPAC names provided, draw the corresponding structure.(a) 2,4-dicyclopropyl-2-ethoxyhexane(b) 1,2-dichloro-1-(2-methylpropyl)-4-nitrocyclohexane(c) 1,3-dicyclopentyl-1,2,3,4-tetramethoxycyclooctane(d) 1-cyclobutyl-4-(1,1-dimethylethyl)-2,4-dinitrononane(e) 1-(1,1-dimethylbutyl)-2-ethoxy-1,2,3-trinitrocyclobutane(f) 1,2,4-tricyclopropyl-1-(2,2-dichloropentyl)cyclohexane(g) 4-(2-chloro-1-methoxyethyl)-1,1-dinitroheptane(h) 3,3,4-trichloro-1-cyclohexoxy-4-(1,1-dichloroethyl)decanearrow_forwardUsing cyclohexane as your starting material, show how you would synthesize each of the following compounds. (Onceyou have shown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.)(a) bromocyclohexane (b) cyclohexenearrow_forward
- 6. Describe concisely a chemical test to distinguish between the following pairs of compounds. (a) n-pentanol and 3-methylpentan-3-ol (b) Ethanal dan pentanal (c) Phenol and benzoic acidarrow_forwardWhich of (a)-(d) is not aromatic? (B) (A) (C) (D)arrow_forwardDraw the structure of the following compounds all showing C and H atoms.(a) 2-methyl -3-iso propyl heptanes(b) Dicyclopropyl methane.arrow_forward
- Question 3 (a) Indicate the following alkenes in order of increasing stability. Justify your answers. (a) (b) (c) (d) (e)arrow_forwardProvide the IUPAC name for each of the following compounds. Pay close attention to stereochemistry. (a) (b) -NH2 (c) OH ОН NH2 NH2 НО .CI Br. NH2 O,N"arrow_forwardProvide IUPAC names for the following compounds.(a) (CH3)2CHCH2CH3 (b) CH3¬C(CH3)2¬CH3arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





