
Concept explainers
(a)
Interpretation:Each stereocenter in the indicated cyclohexane should be labeled as either R or S.
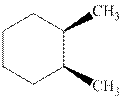
Concept introduction:In order to assign absolute configuration of R and S, Cahn − Ingold − Prelog rules are used and the first step is to assign the priority order on the basis of
The Fischer projection is written along with the priorities assigned and groups are interchanged between adjacent places so as to obtain lowest priority group at the bottom or lowest priority.
If the groups now are arranged are read from highest towards least in clockwise fashion the R is assigned to the stereocenter, if the rotation is anticlockwise the S is assigned at the configuration.
(b)
Interpretation:Each stereocenter in the indicated cyclohexane should be labeled as either R or S.
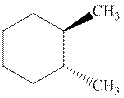
Concept introduction:In order to assign absolute configuration of R and S, Cahn − Ingold − Prelog rules are used and the first step is to assign the priority order on the basis of atomic number. The one with highest atomic number gest highest priority and is designated as a and so on.
The Fischer projection is written along with the priorities assigned and groups are interchanged between adjacent places so as to obtain lowest priority group at the bottom or lowest priority.
If the groups now are arranged are read from highest towards least in clockwise fashion the R is assigned to the stereocenter, if the rotation is anticlockwise the S is assigned at the configuration.
(c)
Interpretation:Each stereocenter in the indicated cyclohexane should be labeled as either R or S.
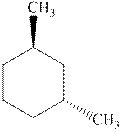
Concept introduction:In order to assign absolute configuration of R and S, Cahn − Ingold − Prelog rules are used and the first step is to assign the priority order on the basis of atomic number. The one with highest atomic number gest highest priority and is designated as a and so on.
The Fischer projection is written along with the priorities assigned and groups are interchanged between adjacent places so as to obtain lowest priority group at the bottom or lowest priority.
If the groups now are arranged are read from highest towards least in clockwise fashion the R is assigned to the stereocenter, if the rotation is anticlockwise the S is assigned at the configuration.
(d)
Interpretation:Each stereocenter in the indicated cyclohexane should be labeled as either R or S.

Concept introduction:In order to assign absolute configuration of R and S, Cahn − Ingold − Prelog rules are used and the first step is to assign the priority order on the basis of atomic number. The one with highest atomic number gest highest priority and is designated as a and so on.
The Fischer projection is written along with the priorities assigned and groups are interchanged between adjacent places so as to obtain lowest priority group at the bottom or lowest priority.
If the groups now are arranged are read from highest towards least in clockwise fashion the R is assigned to the stereocenter, if the rotation is anticlockwise the S is assigned at the configuration.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: Structure and Function
- 5. Draw the structure of (S)-1-bromo-1-chloropropane. Take care to indicate the three- dimensional stereochemistry properly. 6. How many chiral centers does the following compound contain? CH3 7. Label each chiral carbon in the molecule below as having R or S configuration. HO₂C H F. H3C H CH₂CH3arrow_forwardPart D. Do the two structures A and B of each pair drawn below represent the same molecule, constitutional isomers, or stereoisomers? If A and B are stereoisomers, further classify them as enantiomers or diastereomers. H3C Structure A H3C HO H3C H3C Br T H H3C H HO H3CC H C Br HC H H H H CH₂Br CH₂CH3 H CH₂CH3 CH₂OH OH H HO G G H HO H HO G CH3 CH3 CH3 Br Hallo H H3C H Structure B H3C CH3 Br WING H Br HOCH₂ CH3 CH3 HO H CH₂OH H CH₂CH3 H CH₂CH3 H H H H H H Gim CH3 H OH "H ·GII. CH3 HO. OH OH CH3 CH3 OH Relationship of A and Barrow_forwarda. Draw the 14 constitutional isomers of molecular formula C8H9Cl that contain a benzene ring. b.Name all compounds that contain a trisubstituted benzene ring. c.For which compound(s) are stereoisomers possible? Draw all possible stereoisomers.arrow_forward
- 1a. How many stereogenic centers are present 1c. Draw a three-dimensional structure of a in the structure below? Indicate them with asterisk(s). How many stereoisomers stereoisomers are possible? chiral compound with the molecular formula of C4H4Cl₂ that does not have a stereogenic carbon. In addition, draw the enantiomer of this compound. Number of stereogenic centers: Number of stereoisomers possible: 1b. Draw one of the two most stable stereoisomers of the compound in 1a using a planar structure with wedges and dashes. Now draw it in its preferred chair conformation. 1d. Draw two meso compounds with the molecular formula of C7H14.arrow_forward1. Draw all of the stereoisomers of 2,4-dichloropentane. Indicate the relationship between these stereoisomers, such as enantiomers, diastereomers, or meso compounds.arrow_forwardNeed help in E and F. Are stereogenic centers suppose to have 4 different molecules surround them?arrow_forward
- 2. Assign the stereochemical configuration of the selected carbon-carbon double bonds (E, Z or N (not a stereocenter)) that are indicated by the arrows. a) HN H,, H3CO HO H Harrow_forward1a. How many stereogenic centers are present in the structure below? Indicate them with asterisk(s). How many stereoisomers are possible? # of stereogenic centers: # of possible stereoisomers:arrow_forwardDefine each stereocenter as either (R) or (S). If the molecule does not have a stereocenter select “achiral". H OH, H. CH2CH3 H3CH H3CH2C `CH3 HO CH2CH3 CH3 ОН CH2CH3arrow_forward
- 2. Circle all of the stereocenters in the molecules below. If there are none, write "None." a. Adrenaline b. Aspirin ОН c. Limonene d. Testosterone OH OH OH OH H totsarrow_forwardHO CH3 a. Label with asterisks (*) all of the stereocentres in the molecule shown above. b. How many stereoisomers are possible? c. Draw the structures of one pair of enantiomers for the above molecule. (Hint: You should draw two structures using wedge and dashed wedge bonds.)arrow_forward1. How are each of the structures A-F related to structure X? They could be E (enantiomers), D (diastereomers), C (conformers), or I (identical). CH3 CH3 НО. CI- Br CH2OH CI CH3 CI CI Br HOH2C ОН HO Br 'CI HOH,C" OH Br CI HOH2Ċ CI X А. В C CI CI HO CI CI Br CH3 CI Br -CH3 H3C CH2OH ОН Br CI- ČH2OH HO "CH,OH D F Earrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
