
Concept explainers
Interpretation:The chiral molecule should be circled and chiral center should be marked with asterisk along with R and S designation.
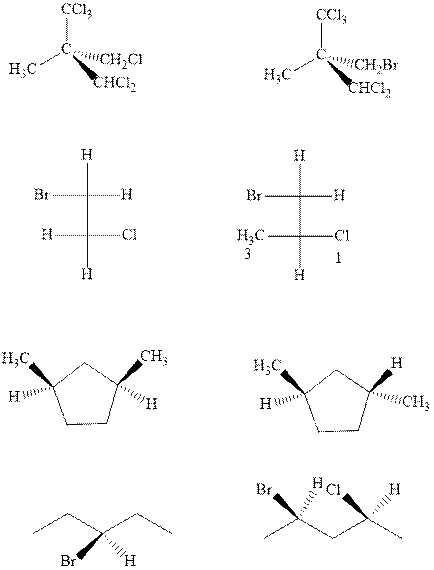
Concept introduction:Chiral carbon is any stereocenter attached to four different alkyl substituents. It is also known as stereocenter. If any two of the substituent happen to be similar the center is regarded as achiral. However, the most essential criteria that help to distinguish a chiral or non-chiral system ispresence of any symmetry element.
In order to assign absolute configuration of R and S, Cahn − Ingold − Prelog rules are used and the first step is to assign the priority order on the basis of
If the groups now are arranged are read from highest towards least in clockwise fashion then R is assigned to the stereocenter if the rotation is anticlockwise then S is assigned at the configuration.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: Structure and Function
- Shown below is Streptomycin, and Neomycin B. Circle and label as many functional groups in these molecules as you can. a. Label each chiral carbon in Streptomycin. How many total stereoisomers exist for Streptomycin? b. Label each chiral carbon in Neomycin B. How many total stereoisomers exist for Neomycin B?arrow_forward10. Draw the enantiomer of the following molecule. Circle each chiral carbon in the original molecule. a. HO Но H- H- FHO- H- HO- HO,arrow_forwardDraw a small circle around the chiral centers in the following molecule. NH2 Brarrow_forward
- Circle the molecules that are chiral. CI H3C OH CH3arrow_forwardShown below is Streptomycin, an antibiotic medication used to treat a number of bacterial infections, including tuberculosis, plague, and endocarditis. Neomycin B has broad-spectrum antibacterial activity. Circle and label as many functional groups in these molecules as you can. Label each chiral carbon in Streptomycin. How many total stereoisomers exist for Streptomycin? Label each chiral carbon in Neomycin B. How many total stereoisomers exist for Neomycin B?arrow_forward2) Circle the asymmetric, chiral carbons. H3C H3C HS H3C Нarrow_forward
- Mark all chiral carbons with a star in the following molecules.arrow_forward15. Naming Stereoisomers with Two Chiral Carbons Using the RS System The (RR) isomer of methyphenidate (Ritalin) is used to treat attention deficit hyperactivity disorder (ADHD). The (S.S) isomer is an antidepressant. Identify the two chiral carbons in the structure below. Is this the (R.R) or the (S.S) isomer? Draw the other isomer. HN- H. ..arrow_forwardCircle and name at least three functional groups in the molecule below. Place an asterisk(*) beside any chiral carbonsarrow_forward
- 5. Assign R and S to each chiral center. Br H. CI NH2 H, OH H... OCH3 CH3 H. NC A A [ [ Choose ] [ Choose ] [ Choose ] C [ [ Choose ] Darrow_forwardThis is one enantiomer of the molecule. OH H CCH2-CH2-CH3 CH3 Draw the structure of the other enantiomer using wedges and dashes. Click and drag to start drawing a structure.arrow_forwarda-Draw the chair structure of the compound. b-Draw the ring-flipped structure of the compound. c-ldentify which of these structures is the most stable or if both are equal in energy explaining the reasoning. a-Draw the chair structure of the compound. b-Draw the ring-flipped structure of the compound. c-Identify which of these structures is the most stable or if both are equal in energy explaining the reasoning.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
