
(a)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
The systematic naming of organic compound is given by
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
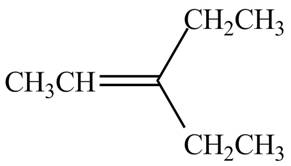
The synthesis of the given compound is shown below.
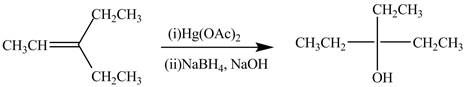
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 1
The alkene
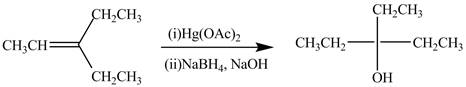
Figure 2
The preparation of the given compound from the alkene and other reagents is shown in Figure 2.
(b)
Interpretation:
The preparation of the given compound from the alkene with and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
![]()
The synthesis of the given compound is shown below.
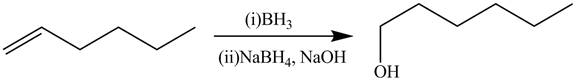
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.
![]()
Figure 3
The alkene
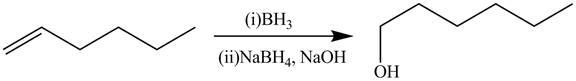
Figure 4
The preparation of the given compound from the alkene and other reagents is shown in Figure 4.
(c)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
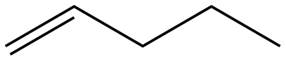
The synthesis of the given compound is shown below.
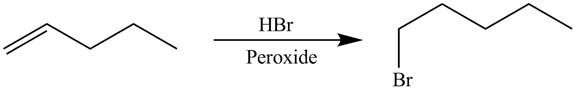
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 5
The alkene

Figure 6
The preparation of the given compound from the alkene and other reagents is shown in Figure 6.
(d)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
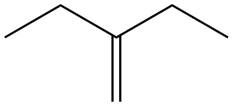
The synthesis of the given compound is shown below.
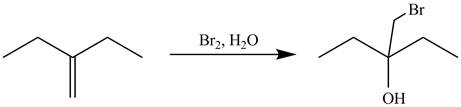
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.
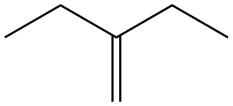
Figure 7
The alkene
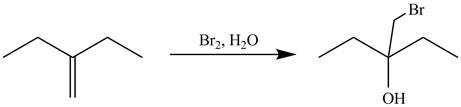
Figure 8
The preparation of the given compound from the alkene and other reagents is shown in Figure 8.
(e)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
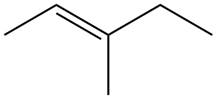
The synthesis of the given compound is shown below.
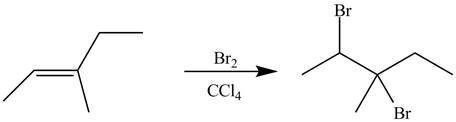
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 9
The alkene
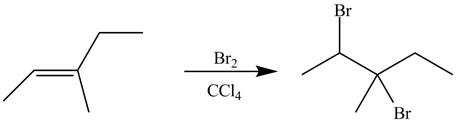
Figure 10
The preparation of the given compound from the alkene and other reagents is shown in Figure 10.
(f)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
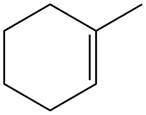
The synthesis of the given compound is shown below.
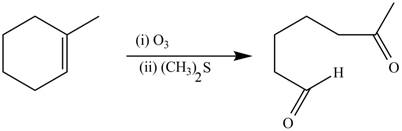
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.
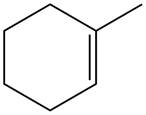
Figure 11
The alkene

Figure 12
The preparation of the given compound from the alkene and other reagents is shown in Figure 12.
(g)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
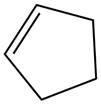
The synthesis of the given compound is shown below.
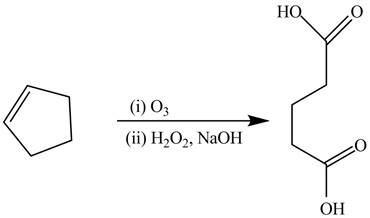
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 13
The alkene cyclopentene undergoes ozonolysis reaction followed by oxidation reaction in presence of
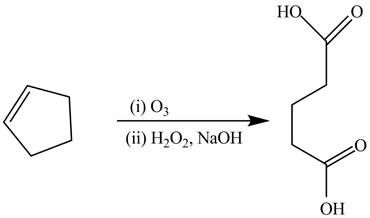
Figure 14
The preparation of the given compound from the alkene and other reagents is shown in Figure 14.
(h)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
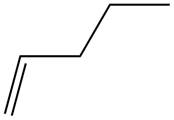
The synthesis of the given compound is shown below.
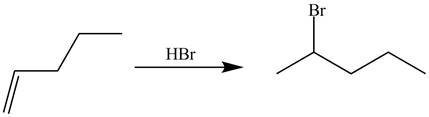
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.
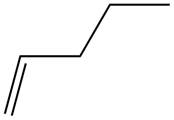
Figure 15
The alkene

Figure 16
The preparation of the given compound from the alkene and other reagentsis shown in Figure 16.
(i)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
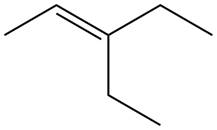
The synthesis of the given compound is shown below.
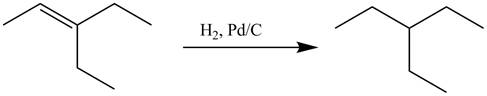
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 17
The alkene
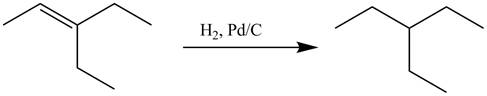
Figure 18
The preparation of the given compound from the alkene and other reagents is shown in Figure 18.
Want to see more full solutions like this?
Chapter 5 Solutions
Organic Chemistry
- (a) Draw the structure of the following :(i) p-Methylbenzaldehyde (ii) 4-Methylpent-3-en-2-one(b) Give chemical tests to distinguish between the following pairs of compounds :(i) Benzoic acid and Ethyl benzoate, (ii) Benzaldehyde and Acetophenone.(iii) Phenol and Benzoic acid.arrow_forwardDescribe how would you distinguish the following pairs, (a) Benzene and cyclohexane (b) Phenol and toluene (c) Phenol and benzoic acid (d) methanol and isopropyl alcoholarrow_forwardShow the chemical reaction on how to convert cyclopentene into these compounds. (a) 1,2-dimethylcyclopropane (b) Cyclopentanol (c) Iodocyclopentane (d) Cyclopentane.arrow_forward
- Biphenyl has the following structure.(a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon?(b) How many pi electrons are there in the two aromatic rings of biphenyl? How does this number compare with that for naphthalene?(c) The heat of hydrogenation for biphenyl is about 418 kJ>mol (100 kcal>mol). Calculate theresonance energy of biphenyl.(d) Compare the resonance energy of biphenyl with that of naphthalene and with that of two benzene rings. Explain thedifference in the resonance energies of naphthalene and biphenyl.arrow_forwardChlorination of 2-butanone yields two isomeric products, each having the molecular formula C4H7ClO. (a) What are these two compounds? (b) Write structural formulas for the enol intermediates that lead to each of these compounds. (c) Using curved arrows, show the flow of electrons in the reaction of each of the enols with Cl2.arrow_forward(a) Give a seperate chemical test to distinguish between the following pairs of compounds:(i) Ethanol and Phenol (ii) 2-Pentanol and 3-Pentanol(b) Explain Kolbe’s reaction with the help of suitable example.arrow_forward
- (a) Compound D undergoes a reaction with hydrogen bromide, HBr to produce 2-bromobutane. D exists as cis-trans isomers and decolourises bromine solution in methylene chloride, CH2CI2. (i) Draw and name the structure of compound D. (ii) Draw two (2) constitutional isomers of compound D.arrow_forwardWhich of the following is true for the reactions of alkyl halides? (a) The characteristic reactions of alkyl halides are oxidation and reduction.(b) The characteristic reactions of alkyl halides are elimination and substitutionc. The characteristic reactions of alkyl halides are addition and substitutiond. Characteristic reactions of alkyl halides are addition and elimination.arrow_forwardArrange the following compounds in increasing order of their property as indicated :(i) CH3COCH3, C6H5COCH3, CH3CHO(reactivity towards nucleophilic addition reaction)(ii) Cl—CH2—COOH, F—CH2—COOH, CH3—COOH (acidic character)arrow_forward
- Give reasons for the following :(i) Phenol is more acidic than methanol.(ii) The C—O—H bond angle in alcohols is slightly less than the tetrahedral angle (190°28′).(iii) (CH3)3C—O—CH3 on reaction with HI gives (CH3)3C—I and CH3—OH as the main products and not (CH3)3C—OH and CH3—I.arrow_forwardand (ii) KMnO4 in aqueous NaOH: 1. Name the following alkenes, and predict the products of their reaction with (i) KMnO4 in aqueous acid (a) (b) 2. Draw structures corresponding to the following IUPAC names: (a) 3-Ethylhept-1-yne (b) 3,5-Dimethylhex-4-en-1-yne (c) Hepta-1,5-diyne (d) 1-Methylcyclopenta-1,3-diene (1) 3. Draw and name all the possible pentyne isomers, C5H8. H H Ç-H [ Pent-1-yne] (2) 4. Predict the products of the following ring is inert to all the indicated reagents.) 4 H 4- (3) 4-C C = C-4 [3-methylbut -1-4 [Pent-2-yhdioselectivity where relevant. (The aromatic CH=CH2 Styrene (a) Styrene+H2 Pd, ? (c) Styrene+HBr ? (b) Styrene+Br2 → ? (d) Styrene+KMnO4 NaOH, H2O ? 5. Suggest structures for alkenes that give the following reaction products. There may be more than one answer for some cases. HyPd catalyst (a) ? Br₂ in CH2Cl2 (b) ? HBr (c) ? 2-Methylhexane 2,3-Dibromo-5-methylhexane 2-Bromo-3-methylheptane CH3 HO OH CH3CHCH2CHCHCH2CH3 KMnO4OH (d) ? H₂Oarrow_forwardStarting from bromoethane, the formation of which of the following compound requires more than one step of reaction? 2 (a) Methoxyethane (b) Ethanol (c) Ethanoic acid (d) Ethenearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





