
Radioactive wastes are packed in a long, thin-walledcylindrical container. The wastes generate thermal energynonuniformly according to the relation
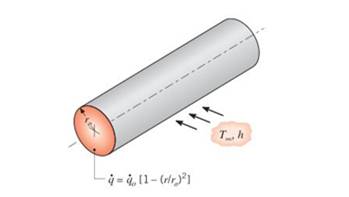
Obtain an expression for the total rate at which energyis generated in a unit length of the container. Use this result to obtain an expression the temperature
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Fundamentals of Heat and Mass Transfer
- number 1 A food product containing 75% moisture content is being frozen. Estimate the specific heat of the product at -10° C when 85% of the water is frozen. The specific heat of the dry product is 2 kJ / (kg ° C). It is assumed that the specific heat of water at -10 ° C is the same as the specific heat of water at 0 ° C, and that the specific heat of ice follows the function Cp es = 0.0062 T Frozen + 2.0649. Cp of frozen product = kJ / kg ° C.arrow_forwardAn open system is often referred to as control volume, which is a properly selected region in space in which mass and energy can flow across the boundaries as figure 1.2. The boundary of a open thermodynamic system is called the control surface Across the Boundaries E = Yes F 0 = Yes w =Yes Control surface ass YES W CONTROL VOLUME energy YES Figure 1.2. A cooling/heating radiator is an example of such a system – give two more examples of such a system.arrow_forwardThe Helmholtz free energy of a liquid column that rises, due to surface tension, inside a capillary tube (Figure 1), as a function of the height, h, is given by: F(h) =(ro)gr^2h^2/2 = -2 pi sigma r h cos teta, where r is the radius of the tube, g is the local ac- celaration of gravity, is the surface tension of the liquid, and is the contact angle of the liquid in contact with the wall of the tube. (a) From this expression, obtain the value of h as a function of the other physical parameters at equilibrium.Calculate the height that water will rise in a capil-lary of diameter 0.05mm. Assume that the contactangle between the water and the tube is zero. Thesurface tension of water at experimental conditionsisσ= 7.73×10−2N/m, and the local accelarationof gravity isg= 9.7m/s2.arrow_forward
- Suppose that we have a wire (or a thin metal rod) of length that is insulated except at the endpoints. Let L denote the position along the wire and let t denote time. The 1- D heat equation is * temperature u 3 insulation O (a^2 u)/(at^2)=c^2 (a^2 u)/(ax^2) (a^2 u)/(at^2 )=c^2 du/əx au/Ət=c^2 (a^2 u)/(ðx^2 ) au/at=c^2 au/Əx nonearrow_forwarda. True/False Given 1-D steady-state conduction through a wall with uniform heat generation, the heat transfer (Q. W) is constant throughout the wall. TRUE FALSE b. True/False Consider convection heat transfer acting on an object that is in contact with a fluid at temperature T-. Ifh (the convection coefficient) gets very large. it tends to drive the surface temperature of that object to the fluid temperature T-. TRUE FALSE c. Short answer You need to calculate the temperature at the free end (tip) of a long fin. The boundary condītion you should NOT use is: Circle one! Convective tip Adiabatic tip Infinite finarrow_forwardFive moles of gas initially at a pressure of 2.00 atm and a volume of 0.300 L has internal energy equal to 91.0 J. In its final state, the gas is at a pressure of 1.50 atm and a volume of 0.800 L, and its internal energy equals 182 J. P (atm) 2.00 B 1.50 V (liters) 0.300 0.800 (a) For the paths IAF, IBF, and IF in the figure above, calculate the work done on the gas. WIAF= W IBF= WIF = (b) For the paths IAF, IBF, and IF in the figure above, calculate the net energy transferred to the gas by heat in the process. QIAF= QIBF=| Need Help? Read Itarrow_forward
- EXPERIMENT PROBLEM: A certain mechanical device with a mechanism similar to a piston contains a certain amount of gas. Upon compression from 55 L to 15 L, the gas released 350 J of heat. Experiment shows that the external pressure of the mechanical device is given by: Pext = AsinV + BcosV + CsinhV + DcoshV Where: A = 100 B = 80 C = 1x10⁻²⁵ D = 5x10⁻³⁰ sin = sine function cos = cosine function sinh = hyperbolic sine function cosh = hyperbolic cosine function V = in liters Pext = in atm Determine the change in internal energy of the system in kilojoules. TIP: usually, expressions involving trigonometric functions are in radians.arrow_forwardEquations of Polytropic Process Definition, diagrams, equations, & sample problems Test Your Skills 14.1 1. A substance undergoes a process such that pV13 = C. If during this process the volume increases by 25% and the initial temperature is 300 K, find the final temperature in K. Your answer Submit Previous activity Jumn to 合曲 60000arrow_forwardIn an electrical discharge machining process, the breakdown voltage across inter electrode gap (IEG) is 200 V and the capacitance of the RC circuit is 50 uF. The energy (in J) released per spark across the IEG is 1arrow_forward
- An electric heater producing 260 W of heat is used to warm up a room containing 7 m3 of air. If we assume the room is perfectly sealed and there is no heat loss through the room boundaries, such that all of the heater output goes into increasing the air temperature, how long will it take to heat up the air in the room from 5.0 °C to 24.1 °C? Give your answer to the nearest minute and assume that the specific volume (v = 0.85 m3/kg) and specific heat capacity at constant volume (cv = 1.005 kJ/(kg K)) remain constant throughout the heating process.arrow_forwardYou witness an industrial fire in which an unknown liquid spilled in to a 8.2 feet diameter circular dike. You were told the tank holding the liquid spilled its entire content of 3,180 kg in to the dike. The fire started and burned for 30 minutes before consuming all the fuel. If the fire had a heat release rate of 50.5 MW what is the chemical heat of combustion of the fuel?arrow_forward3. A metallic rod 20 cm long is heated to a uniform temperature of 100° C. At t = 0 the ends of the bar are plunged into an ice bath at 0° C and thereafter maintained at this temperature. Find an expression for the temperature u(x, t) if the bar is made of cast iron. Material a (cm²/s) Silver 1.71 Copper 1.14 Aluminum 0.86 Cast iron 0.12 Granite 0.011 Brick 0.0038 Water 0.00144 Table 1: Thermal Diffusivity Constants for Common Materialsarrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
