
Using the
a. Determine the order of the group.
b. Verify that the E irreducible representation is orthogonal to each of the other irreduciblerepresentations.
c. For each of the irreducible representations, verify that the sum of the squares of thecharacters equals the order of the group.
d. Reduce the following representations to their component irreducible representations:
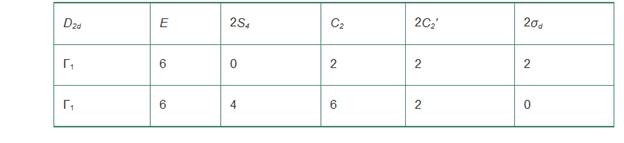
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Inorganic Chemistry
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry (3rd Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: The Central Science (13th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
General Chemistry: Atoms First
- Show that any two of the irreducible representations of the following point groups are orthogonal to each other. a C2 b C2v c D2h d Oh e Tdarrow_forwardShow that any irreducible representation of these point groups is normalized. a C2 b D2d c Oh d C4h e C6varrow_forwardExplain why this proposed irreducible representation for C2v is impossible. EC2A?1100arrow_forward
- In your own words, explain why an object that has more symmetry elements is said to have higher symmetry than an object with fewer symmetry elements.arrow_forward1. A fellow student claims that there is one more irreducible representation for the C2n point group, as follows. C2, Point Group E Ci On I (5) 1 -1 -1 -1 Clearly explain why this student must be incorrect.arrow_forwardA point group has the following sets of elements E, 8 C3, 3C2, 6 S4, 6 od. The order and number of irreducible representations of the group are respectively. ID: AN21340869, NOTE: SCREENSHOT PROHIBITED ID: AN21340869 a. 20 and 10 ID: AN21340869 b. 20 and 6 C ID: AN21340869 c. 24 and 5 ID: AN21340869 d. 24 and 4 First Previous Next Last Questions Bookmarkarrow_forward
- Inorganic chemistry. In the Rules to irreducible representations I can't understand what this part mean. 4. In a given representation (reducible or irreducible), the characters of all matrices belonging to symmetry operations in the same class are identical. can you show me some example of this?arrow_forwardWhat is the point group of NOCl? a. C∞v b. Cs c. none of the above is a correct point group d. D∞h e. C2v D2harrow_forwardHelp me pleasearrow_forward
- 5. Use the character table of the Cav point group to assign the nine "s", "p", and d" orbitals to their corresponding symmetry species (irreducible representation).arrow_forward(4) Determine the point group of CHa and show the symmetry elements/operations using 3D drawing. (5) Determine the point group of SF6 and show the symmetry elements/operations using 3D drawing.arrow_forward1. Consider SF2 molecule, which has non-linear bent geoemtry. What is the point group of this molecule? 1 of 2 u, Draw the normal modes of SF2 using the arrow notation and label them according to the irreducible representations. c) Which of the normal modes are IR active? d) Which of the normal modes are Raman active?arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
