
Determine the point groups for
a. Cyclohexane (chair conformation)
b. Tetrachloroallene
c.
d. A snowflake
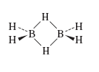
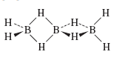
e. Diborane
f. The possible isomers of tribromobenzene
g. A tetrahedron inscribed in a cube in which alternate corners of the cube are also cornersof the tetrahedron.
h.
i. A mountain swallowtail butterfly.
j. The Golden Gate Bridge, in San Francisco, CA
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Inorganic Chemistry
Additional Science Textbook Solutions
Elementary Principles of Chemical Processes, Binder Ready Version
Introduction to Chemistry
Chemistry (7th Edition)
Chemistry
Chemistry
Chemistry & Chemical Reactivity
- Determine if the following species have permanent dipole moments. a Dichloromethane, CH2Cl2 b Chlorobenzene, C6H5Cl c Ammonia, NH3 d Carbon dioxide, CO2.arrow_forwardDetermine the point groups of the following molecules. a Fe(CO)5, which has a trigonal bipyramidal structure, b CO32, which has three resonance structures that contribute to its overall shape, c The perfectly staggered conformation of ethane, d The perfectly eclipsed conformer of ethane.arrow_forwardExplain why a molecule with a center of inversion cannot have a dipole moment.arrow_forward
- Determine the point group of each molecule. Show the different symmetry elements. 7. trans - N₂F2arrow_forwardDraw the three conformations of hydrogen disulfide (H2S2). These are trans (dihedral angle is 180), cis (dihedral angle is 0), and skew (any conformation between the cis and trans). Determine the point group and predict the polarity and chirality for eacharrow_forwardWhich two molecules have the same symmetry point group? a. NH3, BCI3 O b. H20, CH2CI2 c. H20, N02 С. d. PCI3 , ALCI3arrow_forward
- 7. Specify the point group of each of the following species. Br CI P CH3 S. F Fl.. H CI CI CI (C) CI Br F F (A) (В) (D) 8. Examine all the molecules in Question 7 and determine which one is nonpolar and which one has an optical isomer. Why?arrow_forwardAssign the molecular geometry, THREE symmetry elements and the point group for ANY THREE of the following molecules. Molecules Molecular Geometry Symmetry Elements Point Group (Shape) 1. CO 2. CCI, (Not planar) 3. [PtCl,J* (Square planar) 4. NH3 6.arrow_forward4. Describe an object in nature (not a molecule or ion!) whose symmetry corresponds to each of the following point groups, and give a brief explanation why. а. С, b. C2v C. D4harrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
