
Inorganic Chemistry
5th Edition
ISBN: 9780321811059
Author: Gary L. Miessler, Paul J. Fischer, Donald A. Tarr
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
thumb_up100%
Chapter 4, Problem 4.4P
Determine the point groups for
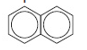
a. Naphthalene
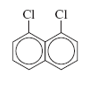
b. 1,8-Dichloronaphthalene
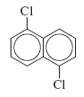
c. 1,5-Dichloronaphthalene
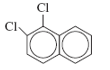
d. 1,2-Dichloronaphthalene
Expert Solution & Answer
Learn your wayIncludes step-by-step video

schedule05:07
Students have asked these similar questions
7.
The following character table (accompanied by a molecule of the corresponding point
group) has some missing information.
E
2 C3
3 C2
?
2 S3
3 oy
X1
1
1
1
1
1
x2+ y?, z2
A2
1
1
-1
1
-1
Rz
E'
2
-1
2
-1
(х, у)
(x2- у?, ху)
A,"
1
X2
1
-1
-1
-1
A2"
1
1
-1
-1
-1
1
X3
BH3
E"
2
-1
-2
1
(Rx, Ry)
(zx, yz)
а.
Based on symmetry labels and Mulliken symbols, what is the missing symmetry element?
b. Provide the missing information in the table:
i. X1 =
ii. X2 =
iii. X3 =
С.
For this point group, what is the symmetry of the following orbitals for an atom located along
the main C3 axis?
i. А Рх
orbital:
ii. A d,? orbital:
iii. A dxy orbital:
What is the point group of NOCl?
a. C∞v
b. Cs
c. none of the above is a correct point group
d. D∞h
e. C2v
f. D2h
Assign the following molecules to point groups: a. HF, b. IF5, c. XeO2F2
Chapter 4 Solutions
Inorganic Chemistry
Ch. 4.1 - Prob. 4.1ECh. 4.1 - Find all the symmetry elements in the following...Ch. 4.2 - Use the procedure described previously to verify...Ch. 4.3 - Prob. 4.4ECh. 4.3 - Verify the transformation matrices for the E and...Ch. 4.3 - Prepare a representation flowchart according to...Ch. 4.4 - Which point groups are possible for chiral...Ch. 4.4 - Write the corresponding 99 transformation matrices...Ch. 4.4 - Using the x, y, and z coordinates for each atom in...Ch. 4.4 - Reduce the following representations to their...
Ch. 4.4 - Prob. 4.11ECh. 4.4 - Analysis of the x, y, and z coordinates of each...Ch. 4.4 - Determine the number of IR-active CO stretching...Ch. 4.4 - Prob. 4.14ECh. 4 - Determine the point groups for a. Ethane...Ch. 4 - Determine the point groups for a. Ethylene b....Ch. 4 - Determine the point groups for a. Acetylene b....Ch. 4 - Determine the point groups for a. Naphthalene b....Ch. 4 - Determine the point groups for a. 1,1’ ...Ch. 4 - Determine the point groups for a. Cyclohexane...Ch. 4 - Determine the point groups for a. A sheet of...Ch. 4 - Determine the point groups for a. A flat oval...Ch. 4 - Determine the point groups for a. A triangular...Ch. 4 - Determine the point groups for the examples of...Ch. 4 - Determine the point groups of the molecules in the...Ch. 4 - Determine the point groups of the molecules and...Ch. 4 - Determine the point groups of the following atomic...Ch. 4 - a. Show that a cube has the same symmetry elements...Ch. 4 - Suppose an octahedron can have either yellow or...Ch. 4 - What point groups are represented by the symbols...Ch. 4 - Prob. 4.17PCh. 4 - Determine the point groups for the following flags...Ch. 4 - Prepare a representation flowchart according to...Ch. 4 - For trans-1,2-dichloroethylene, which has C2h...Ch. 4 - Ethylene has D2h symmetry. a. List all the...Ch. 4 - Using the D2d character table, a. Determine the...Ch. 4 - Reduce the following representations to...Ch. 4 - For D4h symmetry use sketches to show that dxy...Ch. 4 - Prob. 4.25PCh. 4 - XeOF4 has one of the more interesting structures...Ch. 4 - Repeat the procedure from the previous problem,...Ch. 4 - For the following molecules, determine the number...Ch. 4 - Prob. 4.29PCh. 4 - The structure of 1,1,2,2-tetraiododisilane is...Ch. 4 - Both cis and trans isomers of IO2F4 have been...Ch. 4 - White elemental phosphorus consists of tetrahedral...Ch. 4 - Complexes of the general formula Fe(CO)5x( PR3)x...Ch. 4 - Prob. 4.35PCh. 4 - Prob. 4.36PCh. 4 - Prob. 4.37PCh. 4 - Prob. 4.38PCh. 4 - Determine the point groups of the following...Ch. 4 - Prob. 4.40PCh. 4 - Determine the point groups of the following: a....Ch. 4 - Use the Internet to search for molecules with the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Write a balanced chemical equation for each chemical reaction. a. Solid copper reacts with solid sulfur to form...
Introductory Chemistry (6th Edition)
Propose a mechanism for the biosynthesis of -terpineol from geranyl pyrophosphate.
Essential Organic Chemistry (3rd Edition)
If you have an aqueous solution that contains 1.5 moles of HCI, how many moles of ions are in the solution?
a...
Chemistry: The Central Science (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine the point group of the following molecules. a cis1,2 Dichloroethylene b trans1,2 Dichloroethylene c Toluene, C6H5CH3 d 1,3-Cyclohexadiene.arrow_forwardStructural isomers can have very different point groups. Determine the point groups of 1,4cyclohexadiene and 1,3cyclohexadiene, which both have the molecular formula C6H8.arrow_forwardDetermine the point groups of the following molecules. a Fe(CO)5, which has a trigonal bipyramidal structure, b CO32, which has three resonance structures that contribute to its overall shape, c The perfectly staggered conformation of ethane, d The perfectly eclipsed conformer of ethane.arrow_forward
- What is the point group of NOCl? a. C∞v b. Cs c. none of the above is a correct point group d. D∞h e. C2v D2harrow_forward3. Assign each molecule or ion to its proper point group. a. 1,2-dibromoethane (in the gauche conformation) b. 1,2-dibromoethane (in the eclipsed conformation) C. 1-bromo-2-chloro-1,2-difluoroethane (in its most symmetric conformation) d. naphthalenearrow_forward1. Find the molecular geometries and list the symmetry elements of the following molecules and assign the molecules to point groups: a. HF, b. IF5, c. XeO2F2 d. CH4arrow_forward
- . Specify the point group of each of the following species. Br Br F Fl. Fli. Br CI CI F Br CI (A) (В) (C) (D)arrow_forward7. Specify the point group of each of the following species. Br CI F PuCH3 S. F Fl.. F CI CI 'CI Br F (A) (В) (C) (D)arrow_forward3. Assign each molecule or ion to its proper point group. a. NCI3 b. COS CH2N2 (diazomethane) С. d. SOBR2 (thionyl bromide)arrow_forward
- Determine the point group of each molecule. Show the different symmetry elements. 8. cis - N₂F₂arrow_forwardI. Determine the point groups of the following: (found in NH, 1,) D. Tellurium tetrachloride A. Tetrafluoracubane II. Determine the following direct product representations, reducing any reducible ones. 1. On, T1u X Eg D3d, A1g X A2u X Eu 2.arrow_forwardThere are three constitutional isomers of dichlorobenzene: 1. 1,2-dichlorobenzene 2. 1,3-dichlorobenzene 3. 1,4-dichlorobenzene Two of the three isomers belong to the same point group. Which isomer belongs to a different point group than the other two?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,
Group Theory - Learn like Expert with 3D animation | Introduction for Beginners | ONE Chemistry; Author: One Chemistry;https://www.youtube.com/watch?v=Lz2ih8fkgDs;License: Standard YouTube License, CC-BY