
Inorganic Chemistry
5th Edition
ISBN: 9780321811059
Author: Gary L. Miessler, Paul J. Fischer, Donald A. Tarr
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4.4, Problem 4.12E
Analysis of the x, y, and z coordinates of each atom in
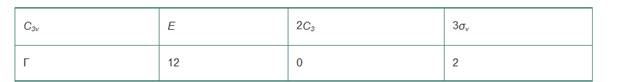
a. Reduce
b. Classify the irreducible representations into translational, rotational, and vibrational modes.
c. Show that the total number of degrees of freedom
d. Which vibrational modes are infrared active?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. In a ro-vibrational spectrum, which branch (P or R) lies at short wavelengths? Explain.
2. The values of B₁ and Bo in a diatomic molecule are different, which is larger? Explain.
3. What causes the differences in peak heights for a rotational spectra.
4. What is meant by centrifugal distortion? How do we correct for it in a rotational spectra?
4. Assume that the characters of a certain reducible representation of the T group are
x(E) = 17, x(C3) = 2, x (C) = 5, x(S4) = - 3, x(0) = - 5 or
T = 17 2 5 -3 - 5. Determine how many times each Irreducible
Representation of T appears in I.
%3D
%3D
5. SAIC Exercise 6.9 How many vibrational modes does an SO, molecule have a) in the plane
of the nuclei, b) perpendicular to the molecular plane? c) Classify them by their symmetry
type.
Determine the symmetries of the
vibrations for SF4 by doing a complete 3N
analysis.
How many infrared-active vibrations do
you expect for SF4?
Character tables are available here.
Chapter 4 Solutions
Inorganic Chemistry
Ch. 4.1 - Prob. 4.1ECh. 4.1 - Find all the symmetry elements in the following...Ch. 4.2 - Use the procedure described previously to verify...Ch. 4.3 - Prob. 4.4ECh. 4.3 - Verify the transformation matrices for the E and...Ch. 4.3 - Prepare a representation flowchart according to...Ch. 4.4 - Which point groups are possible for chiral...Ch. 4.4 - Write the corresponding 99 transformation matrices...Ch. 4.4 - Using the x, y, and z coordinates for each atom in...Ch. 4.4 - Reduce the following representations to their...
Ch. 4.4 - Prob. 4.11ECh. 4.4 - Analysis of the x, y, and z coordinates of each...Ch. 4.4 - Determine the number of IR-active CO stretching...Ch. 4.4 - Prob. 4.14ECh. 4 - Determine the point groups for a. Ethane...Ch. 4 - Determine the point groups for a. Ethylene b....Ch. 4 - Determine the point groups for a. Acetylene b....Ch. 4 - Determine the point groups for a. Naphthalene b....Ch. 4 - Determine the point groups for a. 1,1’ ...Ch. 4 - Determine the point groups for a. Cyclohexane...Ch. 4 - Determine the point groups for a. A sheet of...Ch. 4 - Determine the point groups for a. A flat oval...Ch. 4 - Determine the point groups for a. A triangular...Ch. 4 - Determine the point groups for the examples of...Ch. 4 - Determine the point groups of the molecules in the...Ch. 4 - Determine the point groups of the molecules and...Ch. 4 - Determine the point groups of the following atomic...Ch. 4 - a. Show that a cube has the same symmetry elements...Ch. 4 - Suppose an octahedron can have either yellow or...Ch. 4 - What point groups are represented by the symbols...Ch. 4 - Prob. 4.17PCh. 4 - Determine the point groups for the following flags...Ch. 4 - Prepare a representation flowchart according to...Ch. 4 - For trans-1,2-dichloroethylene, which has C2h...Ch. 4 - Ethylene has D2h symmetry. a. List all the...Ch. 4 - Using the D2d character table, a. Determine the...Ch. 4 - Reduce the following representations to...Ch. 4 - For D4h symmetry use sketches to show that dxy...Ch. 4 - Prob. 4.25PCh. 4 - XeOF4 has one of the more interesting structures...Ch. 4 - Repeat the procedure from the previous problem,...Ch. 4 - For the following molecules, determine the number...Ch. 4 - Prob. 4.29PCh. 4 - The structure of 1,1,2,2-tetraiododisilane is...Ch. 4 - Both cis and trans isomers of IO2F4 have been...Ch. 4 - White elemental phosphorus consists of tetrahedral...Ch. 4 - Complexes of the general formula Fe(CO)5x( PR3)x...Ch. 4 - Prob. 4.35PCh. 4 - Prob. 4.36PCh. 4 - Prob. 4.37PCh. 4 - Prob. 4.38PCh. 4 - Determine the point groups of the following...Ch. 4 - Prob. 4.40PCh. 4 - Determine the point groups of the following: a....Ch. 4 - Use the Internet to search for molecules with the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine the point group of the following molecules. a cis1,2 Dichloroethylene b trans1,2 Dichloroethylene c Toluene, C6H5CH3 d 1,3-Cyclohexadiene.arrow_forwardWhat is the bond order for the lowest excited state of H2+ ? From this single result, propose a general statement about unstable diatomic molecules and bond orders.arrow_forward2. We can form a group of symmetry operations, with our operation being do one operation and do another. For a molecule with an isosceles triangle molecule like water, we talked about the 4 symmetry operations as E (do nothing), C2 (rotation by 180°), σv (the reflection of one hydrogen to the other), and σv’ (the reflection from front to back). By putting coordinates on each of the three atoms (z points along the C2 rotation axis, x points from one hydrogen to the other, and y points out of the plane of the molecule) find the “multiplication” table for this group.arrow_forward
- 4. For each of the following molecules: i. Determine the point group. ii. Determine r (red) as a direct sum of irreducible representations for xyz motion of each atom in the molecule. ii. Determine I (vib) as a direct sum of irreducible representations for molecular vibrations. a. þ,F, b. ХeFsarrow_forwardhow can i get a no. of rotational modes of N2O no. of vibration modes of N2O no. of microwave active modes of N2O no. of infrared active modes of N2Oarrow_forwardList all the electronic transitions possible for a. CH4 b. CH3Cl c. H2C=Oarrow_forward
- For the chlorate anion (CI03-) For the chlorate anion, please give the type of rotational axis and number of mirror planes found in the molecule. O C3, no mirror planes C3, three mirror planes C2, two mirror planes C1, no mirror planesarrow_forwardWhich one isn’t not rotationally raman active? HCN, HF, Br2, CCL4arrow_forwardPerform a vibrational analysis for ammonia. A. How many vibrational modes are possible in total for this non-linear molecule? 6 vibrational modes 4 vibrational modes 2 vibrational modes B. How many of these are Raman active and not IR active? 1 C. Which irreducible representation does the inversion vibration belong to? (What is its Mulliken symbol?) a1 a2 e OOO O00Oarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning
Gerade and Ungerade Molecular Orbitals. (SYMMETRY OF MOLECULAR ORBITALS); Author: Edmerls;https://www.youtube.com/watch?v=dPY-lT5LN60;License: Standard YouTube License, CC-BY
Symmetry and chemical bonding part – 5 Molecular orbital formation (CHE); Author: Vidya-mitra;https://www.youtube.com/watch?v=g-42GmpBu0I;License: Standard Youtube License