
Inorganic Chemistry
5th Edition
ISBN: 9780321811059
Author: Gary L. Miessler, Paul J. Fischer, Donald A. Tarr
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4, Problem 4.5P
Determine the point groups for
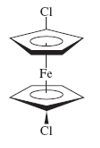
a. 1,1’ − Dichloroferrocene
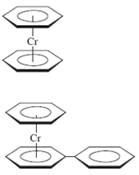
b. Dibenzenechromium (eclipsed conformation)
Expert Solution & Answer
Learn your wayIncludes step-by-step video

schedule12:39
Students have asked these similar questions
I. Determine the point groups of the
following:
(found in NH, 1,)
D. Tellurium tetrachloride
A. Tetrafluoracubane
II. Determine the following direct
product representations, reducing any
reducible ones.
1. On, T1u X Eg
D3d, A1g X A2u X Eu
2.
4. Describe an object in nature (not a molecule or ion!) whose symmetry corresponds to each of
the following point groups, and give a brief explanation why.
а. С,
b. C2v
C.
D4h
Determine the point group of each molecule.
Show the different symmetry elements.
5.S0₂
Chapter 4 Solutions
Inorganic Chemistry
Ch. 4.1 - Prob. 4.1ECh. 4.1 - Find all the symmetry elements in the following...Ch. 4.2 - Use the procedure described previously to verify...Ch. 4.3 - Prob. 4.4ECh. 4.3 - Verify the transformation matrices for the E and...Ch. 4.3 - Prepare a representation flowchart according to...Ch. 4.4 - Which point groups are possible for chiral...Ch. 4.4 - Write the corresponding 99 transformation matrices...Ch. 4.4 - Using the x, y, and z coordinates for each atom in...Ch. 4.4 - Reduce the following representations to their...
Ch. 4.4 - Prob. 4.11ECh. 4.4 - Analysis of the x, y, and z coordinates of each...Ch. 4.4 - Determine the number of IR-active CO stretching...Ch. 4.4 - Prob. 4.14ECh. 4 - Determine the point groups for a. Ethane...Ch. 4 - Determine the point groups for a. Ethylene b....Ch. 4 - Determine the point groups for a. Acetylene b....Ch. 4 - Determine the point groups for a. Naphthalene b....Ch. 4 - Determine the point groups for a. 1,1’ ...Ch. 4 - Determine the point groups for a. Cyclohexane...Ch. 4 - Determine the point groups for a. A sheet of...Ch. 4 - Determine the point groups for a. A flat oval...Ch. 4 - Determine the point groups for a. A triangular...Ch. 4 - Determine the point groups for the examples of...Ch. 4 - Determine the point groups of the molecules in the...Ch. 4 - Determine the point groups of the molecules and...Ch. 4 - Determine the point groups of the following atomic...Ch. 4 - a. Show that a cube has the same symmetry elements...Ch. 4 - Suppose an octahedron can have either yellow or...Ch. 4 - What point groups are represented by the symbols...Ch. 4 - Prob. 4.17PCh. 4 - Determine the point groups for the following flags...Ch. 4 - Prepare a representation flowchart according to...Ch. 4 - For trans-1,2-dichloroethylene, which has C2h...Ch. 4 - Ethylene has D2h symmetry. a. List all the...Ch. 4 - Using the D2d character table, a. Determine the...Ch. 4 - Reduce the following representations to...Ch. 4 - For D4h symmetry use sketches to show that dxy...Ch. 4 - Prob. 4.25PCh. 4 - XeOF4 has one of the more interesting structures...Ch. 4 - Repeat the procedure from the previous problem,...Ch. 4 - For the following molecules, determine the number...Ch. 4 - Prob. 4.29PCh. 4 - The structure of 1,1,2,2-tetraiododisilane is...Ch. 4 - Both cis and trans isomers of IO2F4 have been...Ch. 4 - White elemental phosphorus consists of tetrahedral...Ch. 4 - Complexes of the general formula Fe(CO)5x( PR3)x...Ch. 4 - Prob. 4.35PCh. 4 - Prob. 4.36PCh. 4 - Prob. 4.37PCh. 4 - Prob. 4.38PCh. 4 - Determine the point groups of the following...Ch. 4 - Prob. 4.40PCh. 4 - Determine the point groups of the following: a....Ch. 4 - Use the Internet to search for molecules with the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
For the generic equilibrium HA(aq) ⇌ H + (aq) + A- (aq), which of these statements is true?
The equilibrium con...
Chemistry: The Central Science (14th Edition)
23. Give the symbol and name for (a) an isotope with a mass number of 37 and an atomic number of 17 and (b) an ...
Chemistry For Changing Times (14th Edition)
a. Draw the most stable conformation of pentane for rotation about the C-2C-3 bond. b. Draw the least stable co...
Essential Organic Chemistry (3rd Edition)
List these three types of radiationinfrared, X-ray, and radio wavesin order of: a. increasing energy per photon...
Introductory Chemistry (5th Edition) (Standalone Book)
The glycine cleavage system is a group of four enzymes that together catalyze the following reaction: glycine+T...
Organic Chemistry
Use electronegativities to predict the direction of the dipole moments of the following bonds. a. CCl b. CO c. ...
Organic Chemistry (9th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine the point group of the following molecules. a cis1,2 Dichloroethylene b trans1,2 Dichloroethylene c Toluene, C6H5CH3 d 1,3-Cyclohexadiene.arrow_forwardStructural isomers can have very different point groups. There are three isomers of dichlorobenzene. Identify the point groups of the three isomers.arrow_forwardStructural isomers can have very different point groups. Determine the point groups of 1,4cyclohexadiene and 1,3cyclohexadiene, which both have the molecular formula C6H8.arrow_forward
- Determine the point groups of the following molecules. a Hydrogen selenide, H2Se b Partially deuterated hydrogen sulfide, or HDS c The chair conformer of cyclohexane, C6H12 d The boat conformer of cyclohexane, C6H12arrow_forwardFigure 13.27 shows the structure of the molecule porphine. Figure 13.27 The structure of porphine. Determine the symmetry elements present in the molecule, and its point group. Does the point group change if an Fe2+ ion is substituted for the two hydrogen atoms in the center of the porphine ring?arrow_forward7. The following character table (accompanied by a molecule of the corresponding point group) has some missing information. E 2 C3 3 C2 ? 2 S3 3 oy X1 1 1 1 1 1 x2+ y?, z2 A2 1 1 -1 1 -1 Rz E' 2 -1 2 -1 (х, у) (x2- у?, ху) A," 1 X2 1 -1 -1 -1 A2" 1 1 -1 -1 -1 1 X3 BH3 E" 2 -1 -2 1 (Rx, Ry) (zx, yz) а. Based on symmetry labels and Mulliken symbols, what is the missing symmetry element? b. Provide the missing information in the table: i. X1 = ii. X2 = iii. X3 = С. For this point group, what is the symmetry of the following orbitals for an atom located along the main C3 axis? i. А Рх orbital: ii. A d,? orbital: iii. A dxy orbital:arrow_forward
- 3. Assign each molecule or ion to its proper point group. a. cyclopropylene b. aziridine (hint: be very mindful of lone pairs) c. NbF,2-arrow_forwardDetermine the point groups for H -C = C - Ph (Ph = phenyl)arrow_forwardDraw a Lewis structure and determine the point group for each of the following molecules. Indicate formal charges for ions. Indicate 3D stereochemistry by drawing solid and hashed bonds. a. XeO3 b. SOF4 c. ClOF2+ d. XeO2F2 e. TeF42-arrow_forward
- Assign the following molecules to point groups: a. HF, b. IF5, c. XeO2F2arrow_forwardAssign a point group to each of the following compounds and identify how the Pz orbital on the central atom transforms under that respective symmetry. a. NH2Cl b. BF4 - c. HCNarrow_forwardDetermine the point group of each molecule. Show the different symmetry elements. 2. BHAarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,
Group Theory - Learn like Expert with 3D animation | Introduction for Beginners | ONE Chemistry; Author: One Chemistry;https://www.youtube.com/watch?v=Lz2ih8fkgDs;License: Standard YouTube License, CC-BY